Brain Cancer Immunotherapy (Review)
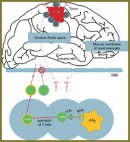
The review analyzes Russian and foreign reports concerned with a rapidly developing brain cancer treatment technique — immunotherapy. There has been presented a current view on the basic concept of antitumor immunity, on the problem of immune system interaction with a tumor in general and under the conditions of an immunologically privileged nervous system, shown the theoretical background of efficiency of immunotherapy used against brain cancer (the capability of tumor antigens and activated lymphocytes to penetrate the blood-brain barrier). There has been demonstrated the role of a transforming growth factor β, interleukin 10, cyclooxygenase-2, prostaglandin Е2, protein MCP-1, interactions Fas-receptor/Fas-ligand, antigen-4 cytotoxic Т-lymphocytes in tumor immunoresistance development. The review presents a current classification of the types of active and passive immunotherapy, each of the types being considered separately specifying the characteristics, the results of preclinical and clinical trials of each type efficiency, and possible side effects. Special attention has been paid to a new concept of a key role of tumor stem cells in the pathogenesis of cerebral gliomas and the target action on these cells.
- Stupp R., Mason W.P., van den Bent M.J., et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005; 352(10): 987–996, http://dx.doi.org/10.1056/NEJMoa043330.
- Olyushin V.E. Cerebral gliomas: a brief review and a treatment protocol. Neyrokhirurgiya 2005; 4: 41–47.
- Konovalov A.N., Potapov A.A., Olyushin V.E., et al. Standards, options and recommendations in the management of primary CNS tumors (2013–2014). Moscow; 2013.
- Dillman R.O. Cancer immunotherapy. Cancer Biother Radiopharm 2011; 26(1): 1–64, http://dx.doi.org/10.1089/cbr.2010.0902.
- Kuramoto T. Detection of MAGE-1 tumor antigen in brain tumor. Kurume Med J 1997; 44(1): 43–51, http://dx.doi.org/10.2739/kurumemedj.44.43.
- Ueda R., Yoshida K., Kawakami Y., et al. Expression of a transcriptional factor, SOX6, in human gliomas. Brain Tumor Pathol 2004; 21(1): 35–38.
- Chi D.D., Merchant R.E., Rand R., et al. Molecular detection of tumor-associated antigens shared by human cutaneous melanomas and gliomas. Am J Pathol 1997; 150(6): 2143–2152.
- Liu G., Ying H., Zeng G., et al. HER-2, gp100, and MAGE-1 are expressed in human glioblastoma and recognized by cytotoxic T cells. Cancer Res 2004; 64(14): 4980–4986, http://dx.doi.org/10.1158/0008-5472.CAN-03-3504.
- Heimberger A.B., Crotty L.E., Archer G.E., et al. Epidermal growth factor receptor VIII peptide vaccination is efficacious against established intracerebral tumors. Clin Cancer Res 2003; 9(11): 4247–4254.
- Okano F., Storkus W.J., Chambers W.H., et al. Identification of a novel HLA-A*0201-restricted, cytotoxic T lymphocyte epitope in a human glioma-associated antigen, interleukin 13 receptor alpha2 chain. Clin Cancer Res 2002; 8(9): 2851–2855.
- Hatano M., Eguchi J., Tatsumi T., et al. EphA2 as a glioma-associated antigen: a novel target for glioma vaccines. Neoplasia 2005; 7(8): 717–722.
- Jin M., Komohara Y., Shichijo S., et al. Identification of EphB6 variant-derived epitope peptides recognized by cytotoxic T-lymphocytes from HLA-A24+ malignant glioma patients. Oncol Rep 2008; 19(5): 1277–1283, http://dx.doi.org/10.3892/or.19.5.1277.
- Liu G., Yu J.S., Zeng G., et al. AIM-2: a novel tumor antigen is expressed and presented by human glioma cells. J Immunother 2004; 27(3): 220–226.
- Hashiba T., Izumoto S., Kagawa N., et al. Expression of WT1 protein and correlation with cellular proliferation in glial tumors. Neurol Med Chir (Tokyo) 2007; 47(4): 165–170; discussion 170, http://dx.doi.org/10.2176/nmc.47.165.
- Nonaka Y., Tsuda N., Shichijo S., et al. Recognition of ADP-ribosylation factor 4-like by HLA-A2-restricted and tumor-reactive cytotoxic T lymphocytes from patients with brain tumors. Tissue Antigens 2002; 60(4): 319–327, http://dx.doi.org/10.1034/j.1399-0039.2002.600406.x.
- Murayama K., Kobayashi T., Imaizumi T., et al. Expression of the SART3 tumor-rejection antigen in brain tumors and induction of cytotoxic T lymphocytes by its peptides. J Immunother 2000; 23(5): 511–518.
- Schmitz M., Wehner R., Stevanovic S., et al. Identification of a naturally processed T cell epitope derived from the glioma-associated protein SOX11. Cancer Lett 2007; 245(1–2): 331–336, http://dx.doi.org/10.1016/j.canlet.2006.01.014.
- Harada M., Ishihara Y., Itoh K., Yamanaka R. Kinesin superfamily protein-derived peptides with the ability to induce glioma-reactive cytotoxic T lymphocytes in human leukocyte antigen-A24+ glioma patients. Oncol Rep 2007; 17(3): 629–636, http://dx.doi.org/10.3892/or.17.3.629.
- Dunn G.P., Old L.J., Schreiber R.D. The three Es of cancer immunoediting. Annu Rev Immunol 2004; 22: 329–360, http://dx.doi.org/10.1146/annurev.immunol.22.012703.104803.
- Muldoon L.L., Alvarez J.I., Begley D.J., et al. Immunologic privilege in the central nervous system and the blood-brain barrier. J Cereb Blood Flow Metab 2013; 33(1): 13–21, http://dx.doi.org/10.1038/jcbfm.2012.153.
- Chekhonin V.P., Baklaushev V.P., Yusubalieva G.M., et al. Fundamental and application aspects of blood-brain barrier study. Vestnik Rossiyskoy Akademii meditsinskikh nauk 2012; 8: 66–78.
- Bechmann I., Galea I., Perry V.H. What is the blood-brain barrier (not)? Trends Immunol 2007; 28(1): 5–11, http://dx.doi.org/10.1016/j.it.2006.11.007.
- Tambur A.R. Transplantation immunology and the central nervous system. Neurol Res 2004; 26(3): 243–255, http://dx.doi.org/10.1179/016164104225013932.
- Anirban G. Immune connection in glioma: fiction, fact and option, glioma, in glioma — exploring its biology and practical relevance. In: Glioma — exploring its biology and practical relevance. Ed. by Ghosh D.A. InTech; 2011; р. 305–324.
- Hickey W.F. Leukocyte traffic in the central nervous system: the participants and their roles. Semin Immunol 1999; 11(2): 125–137, http://dx.doi.org/10.1006/smim.1999.0168.
- Flügel A., Schwaiger F.W., Neumann H., et al. Neuronal FasL induces cell death of encephalitogenic T lymphocytes. Brain Pathol 2000; 10(3): 353–364.
- Yang I., Han S.J., Kaur G., et al. The role of microglia in central nervous system immunity and glioma immunology. J Clin Neurosci 2010; 17(1): 6–10, http://dx.doi.org/10.1016/j.jocn.2009.05.006.
- Karman J., Ling C., Sandor M., et al. Dendritic cells in the initiation of immune responses against central nervous system-derived antigens. Immunol Lett 2004; 92(1–2): 107–115, http://dx.doi.org/10.1016/j.imlet.2003.10.017.
- Goldmann J.E., Kwidzinski C., Brandt C., et al. T cells traffic from brain to cervical lymph nodes via the cribroid plate and the nasal mucosa. J Leukoc Biol 2006; 80(4): 797–801, http://dx.doi.org/10.1189/jlb.0306176.
- Ransohoff R.M., Kivisakk P., Kidd G. Three or more routes for leukocyte migration into the central nervous system. Nat Rev Immunol 2003; 3(7): 569–581, http://dx.doi.org/10.1038/nri1130.
- Calzascia T., Masson F., Di Berardino-Besson W., et al. Homing phenotypes of tumor-specific CD8 T cells are predetermined at the tumor site by crosspresenting APCs. Immunity 2005; 22(2): 175–184, http://dx.doi.org/10.1016/j.immuni.2004.12.008.
- Okada H., Kohanbash G., Zhu X., et al. Immunotherapeutic approaches for glioma. Crit Rev Immunol 2009; 29(1): 1–42, http://dx.doi.org/10.1615/CritRevImmunol.v29.i1.10.
- Roy L.O., Poirier M.B., Fortin D. Transforming growth factor-beta and its implication in the malignancy of gliomas. Target Oncol 2014; http://dx.doi.org/10.1007/s11523-014-0308-y. [Epub ahead of print].
- Moore K.W., de Waal Malefyt R., Coffman R.L., et al. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol 2001; 19: 683–765, http://dx.doi.org/10.1146/annurev.immunol.19.1.683.
- Jiang J., Dingledine R. Role of prostaglandin receptor EP2 in the regulations of cancer cell proliferation, invasion, and inflammation. J Pharmacol Exp Ther 2013; 344(2): 360–367, http://dx.doi.org/10.1124/jpet.112.200444.
- Zhang J., Sarkar S., Cua R., et al. A dialog between glioma and microglia that promotes tumor invasiveness through the CCL2/CCR2/interleukin-6 axis. Carcinogenesis 2012; 33(2): 312–319, http://dx.doi.org/10.1093/carcin/bgr289.
- Shinohara H., Yagita H., Ikawa Y., Oyaizu N. Fas drives cell cycle progression in glioma cells via extracellular signal-regulated kinase activation. Cancer Res 2000; 60(6): 1766–1772.
- Salama A.K., Hodi F.S. Cytotoxic T-lymphocyte-associated antigen-4. Clin Cancer Res 2011; 17(14): 4622–4628, http://dx.doi.org/10.1158/1078-0432.ccr-10-2232.
- Gabrilovich D.I., Ostrand-Rosenberg S., Bronte V. Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol 2012; 12(4): 253–268. http://dx.doi.org/10.1038/nri3175.
- Sica A., Schioppa T., Mantovani A., et al., Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer 2006; 42(6): 717–727, http://dx.doi.org/10.1016/j.ejca.2006.01.003.
- Ohkura N., Kitagawa Y., Sakaguchi S. Development and maintenance of regulatory T cells. Immunity 2013; 38(3): 414–423, http://dx.doi.org/10.1016/j.immuni.2013.03.002.
- Mirghorbani M., Van Gool S., Rezaei N. Myeloid-derived suppressor cells in glioma. Expert Rev Neurother 2013; 13(12): 1395–1406, http://dx.doi.org/10.1586/14737175.2013.857603.
- Ooi Y.C., Tran P., Ung N., et al. The role of regulatory T-cells in glioma immunology. Clin Neurol Neurosurg 2014; 119: 125–132, http://dx.doi.org/10.1016/j.clineuro.2013.12.004.
- Grauer O.M., Nierkens S., Bennink E., et al. CD4+FoxP3+ regulatory T cells gradually accumulate in gliomas during tumor growth and efficiently suppress antiglioma immune responses in vivo. Int J Cancer 2007; 121(1): 95–105, http://dx.doi.org/10.1002/ijc.22607.
- Gousias K., Markou M., Arzoglou V., et al. Frequent abnormalities of the immune system in gliomas and correlation with the WHO grading system of malignancy. J Neuroimmunol 2010; 226(1–2): 136–142, http://dx.doi.org/10.1016/j.jneuroim.2010.05.027.
- Usatov S.A., Kovalenko A.P., Zallum Kh., et al. The immune status condition in patients with cerebral gliomas. Ukraїns’kiy medichniy al’manakh 2010; 13(1): 158–161.
- Brantley E.C., Benveniste E.N. Signal transducer and activator of transcription-3: a molecular hub for signaling pathways in gliomas. Mol Cancer Res 2008; 6(5): 675–684, http://dx.doi.org/10.1158/1541-7786.MCR-07-2180.
- Dix A.R., Brooks W.H., Roszman T.L., et al. Immune defects observed in patients with primary malignant brain tumors. J Neuroimmunol 1999; 100(1–2): 216–232, http://dx.doi.org/10.1016/S0165-5728(99)00203-9.
- Giometto B., Bozza F., Faresin F., et al. Immune infiltrates and cytokines in gliomas. Acta Neurochir (Wien) 1996; 138(1): 50–56.
- Lang R., Patel D., Morris J.J., et al. Shaping gene expression in activated and resting primary macrophages by IL-10. J Immunol 2002; 169(5): 2253–2263, http://dx.doi.org/10.4049/jimmunol.169.5.2253.
- Mancino A., Lawrence T. Nuclear factor-kappaB and tumor-associated macrophages. Clin Cancer Res 2010; 16(3): 784–789, http://dx.doi.org/10.1158/1078-0432.CCR-09-1015.
- Day E.D., Lassiter S., Woodhall B., et al. The localization of radioantibodies in human brain tumors. I. Preliminary exploration. Cancer Res 1965; 25(6): 773–778.
- Bidros D.S., Vogelbaum M.A. Novel drug delivery strategies in neuro-oncology. Neurotherapeutics 2009; 6(3): 539–546, http://dx.doi.org/10.1016/j.nurt.2009.04.004.
- Liu H.L., Hua M.Y., Chen P.Y., et al. Blood-brain barrier disruption with focused ultrasound enhances delivery of chemotherapeutic drugs for glioblastoma treatment. Radiology 2010; 255(2): 415–425, http://dx.doi.org/10.1148/radiol.10090699.
- Medyanik I.A., Mukhina I.V., Yakovleva E.I., et al. The method of temporary increase of blood-brain barrier penetration. Patent RF No.2391107. 2010.
- Bigner D.D., Brown M., Coleman R.E., et al. Phase I studies of treatment of malignant gliomas and neoplastic meningitis with 131I-radiolabeled monoclonal antibodies anti-tenascin 81C6 and anti-chondroitin proteoglycan sulfate Me1-14 F (ab’)2 — a preliminary report. J Neurooncol 1995; 24(1): 109–122.
- Riva P., Arista A., Franceschi G., et al. Local treatment of malignant gliomas by direct infusion of specific monoclonal antibodies labeled with 131I: comparison of the results obtained in recurrent and newly diagnosed tumors. Cancer Res 1995; 55(23 Suppl): 5952s–5956s.
- Eller J.L., Longo S.L., Hicklin D.J., Canute G.W. Activity of anti-epidermal growth factor receptor monoclonal antibody C225 against glioblastoma multiforme. Neurosurgery 2002; 51(4): 1005–1013; discussion 1013–1014.
- Neyns B., Sadones J., Joosens E., et al. Stratified phase II trial of cetuximab in patients with recurrent high-grade glioma. Ann Oncol 2009; 20(9): 1596–1603, http://dx.doi.org/10.1093/annonc/mdp032.
- Berezowska S., Schlegel J. Targeting ErbB receptors in high-grade glioma. Curr Pharm Des 2011; 17(23): 2468–2487, http://dx.doi.org/10.2174/138161211797249233.
- Vredenburgh J.J., Desjardins A., Herndon J.E. 2nd, et al. Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res 2007; 13(4): 1253–1259, http://dx.doi.org/10.1158/1078-0432.CCR-06-2309.
- Friedman H.S., Prados M.D., Wen P.Y., et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol 2009; 27(28): 4733–4740, http://dx.doi.org/10.1200/JCO.2008.19.8721.
- Cecchi M., Vaiani M., Ceroti M., et al. A retrospective observational analysis to evaluate the off-label use of bevacizumab alone or with irinotecan in recurrent glioblastoma. Int J Clin Pharm 2013; 35(3): 483–487, http://dx.doi.org/10.1007/s11096-013-9765-0.
- Chamberlain M.C., Johnston S.K. Salvage therapy with single agent bevacizumab for recurrent glioblastoma. J Neurooncol 2010; 96(2): 259–269, http://dx.doi.org/10.1007/s11060-009-9957-6.
- Zhang G., Huang S., Wang Z. A meta-analysis of bevacizumab alone and in combination with irinotecan in the treatment of patients with recurrent glioblastoma multiforme. J Clin Neurosci 2012; 19(12): 1636–1640, http://dx.doi.org/10.1016/j.jocn.2011.12.028.
- Curtin J.F., Candolfi M., Fakhouri T.M., et al. Treg depletion inhibits efficacy of cancer immunotherapy: implications for clinical trials. PLoS One 2008; 3(4): e1983, http://dx.doi.org/10.1371/journal.pone.0001983.
- Mitchell D.A., Cui X., Schmittling R.J., et al. Monoclonal antibody blockade of IL-2 receptor alpha during lymphopenia selectively depletes regulatory T cells in mice and humans. Blood 2011; 118(11): 3003–3012, http://dx.doi.org/10.1182/blood-2011-02-334565.
- Blancher A., Roubinet F., Grancher A.S., et al. Local immunotherapy of recurrent glioblastoma multiforme by intracerebral perfusion of interleukin-2 and LAK cells. Eur Cytokine Netw 1993; 4(5): 331–341.
- Boiardi A., Silvani A., Ruffini P.A., et al. Loco-regional immunotherapy with recombinant interleukin-2 and adherent lymphokine-activated killer cells (A-LAK) in recurrent glioblastoma patients. Cancer Immunol Immunother 1994; 39(3): 193–197.
- Hayes R.L., Koslow M., Hiesiger E.M., et al. Improved long term survival after intracavitary interleukin-2 and lymphokine-activated killer cells for adults with recurrent malignant glioma. Cancer 1995; 76(5): 840–852.
- Dillman R.O., Duma C.M., Ellis R.A., et al. Intralesional lymphokine-activated killer cells as adjuvant therapy for primary glioblastoma. J Immunother 2009; 32(9): 914–919, http://dx.doi.org/10.1097/CJI.0b013e3181b2910f.
- Plautz G.E., Barnett G.H., Miller D.W., et al. Systemic T cell adoptive immunotherapy of malignant gliomas. J Neurosurg 1998; 89(1): 42–51, http://dx.doi.org/10.3171/jns.1998.89.1.0042.
- Saied A., Pillarisetty V.G., Katz S.C. Immunotherapy for solid tumors — a review for surgeons. J Surg Res 2014; 187(2): 525–535, http://dx.doi.org/10.1016/j.jss.2013.12.018.
- Yu J.S., Wheeler C.J., Zeltzer P.M., et al. Vaccination of malignant glioma patients with peptide-pulsed dendritic cells elicits systemic cytotoxicity and intracranial T-cell infiltration. Cancer Res 2001; 61(3): 842–847.
- Kikuchi T., Akasaki Y., Irie M., et al. Results of a phase I clinical trial of vaccination of glioma patients with fusions of dendritic and glioma cells. Cancer Immunol Immunother 2001; 50(7): 337–344.
- Ardon H., Van Gool S.W., Verschuere T., et al. Integration of autologous dendritic cell-based immunotherapy in the standard of care treatment for patients with newly diagnosed glioblastoma: results of the HGG-2006 phase I/II trial. Cancer Immunol Immunother 2012; 61(11): 2033–2044, http://dx.doi.org/10.1007/s00262-012-1261-1.
- Kozlov V.A., Chernykh E.R. Current immune therapy problems in oncology. Byulleten’ SO RAMN 2004; 2(112): 13–19.
- Rosenberg S.A., Yang J.C., Restifo N.P. Cancer immunotherapy: moving beyond current vaccines. Nat Med 2004; 10(9): 909–15, http://dx.doi.org/10.1038/nm1100.
- Sampson J.H., Archer G.E., Mitchell D.A., et al. Tumor-specific immunotherapy targeting the EGFRvIII mutation in patients with malignant glioma. Semin Immunol 2008; 20(5): 267–275, http://dx.doi.org/10.1016/j.smim.2008.04.001.
- Sampson J.H., Heimberger A.B., Archer G.E., et al. Immunologic escape after prolonged progression-free survival with epidermal growth factor receptor variant III peptide vaccination in patients with newly diagnosed glioblastoma. J Clin Oncol 2010; 28(31): 4722–4729, http://dx.doi.org/10.1200/JCO.2010.28.6963.
- Yajima N., Yamanaka R., Mine T., et al. Immunologic evaluation of personalized peptide vaccination for patients with advanced malignant glioma. Clin Cancer Res 2005; 11(16): 5900–5911, http://dx.doi.org/10.1158/1078-0432.CCR-05-0559.
- Mine T., Sato Y., Noguchi M., et al. Humoral responses to peptides correlate with overall survival in advanced cancer patients vaccinated with peptides based on pre-existing, peptide-specific cellular responses. Clin Cancer Res 2004; 10(3): 929–937, http://dx.doi.org/10.1158/1078-0432.CCR-1117-3.
- Terasaki M., Shibui S., Narita Y., et al. Phase I trial of a personalized peptide vaccine for patients positive for human leukocyte antigen — A24 with recurrent or progressive glioblastoma multiforme. J Clin Oncol 2011; 29(3): 337–44, http://dx.doi.org/10.1200/JCO.2010.29.7499.
- Sampetrean O., Saya H. Characteristics of glioma stem cells. Brain Tumor Pathol 2013; 30(4): 209–214, http://dx.doi.org/10.1007/s10014-013-0141-5.
- Heywood R.M., Marcus H.J., Ryan D.J., et al. A review of the role of stem cells in the development and treatment of glioma. Acta Neurochir (Wien) 2012; 154(6): 951–969; discussion 969, http://dx.doi.org/10.1007/s00701-012-1338-9.
- Qiu B., Zhang D., Tao J., et al. Human brain glioma stem cells are more invasive than their differentiated progeny cells in vitro. J Clin Neurosci 2012; 19(1): 130–134, http://dx.doi.org/10.1016/j.jocn.2011.06.014.
- Singh S.K., Hawkins C., Clarke I.D., et al. Identification of human brain tumour initiating cells. Nature 2004; 432(7015): 396–401, http://dx.doi.org/10.1038/nature03128.
- Dean M., Fojo T., Bates S. Tumour stem cells and drug resistance. Nat Rev Cancer 2005; 5(4): 275–284, http://dx.doi.org/10.1038/nrc1590.
- Persano L., Rampazzo E., Basso G., et al. Glioblastoma cancer stem cells: role of the microenvironment and therapeutic targeting. Biochem Pharmacol 2013; 85(5): 612–622, http://dx.doi.org/10.1016/j.bcp.2012.10.001.
- Bao S., Wu Q., McLendon R.E., et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006; 444(7120): 756–760, http://dx.doi.org/10.1038/nature05236.
- Huang Z., Cheng L., Guryanova O.A., et al. Cancer stem cells in glioblastoma — molecular signaling and therapeutic targeting. Protein Cell 2010; 1(7): 638–655, http://dx.doi.org/10.1007/s13238-010-0078-y.
- Tate M.C., Aghi M.K. Biology of angiogenesis and invasion in glioma. Neurotherapeutics 2009; 6(3): 447–457, http://dx.doi.org/10.1016/j.nurt.2009.04.001.
- Hovinga K.E., Stalpers L.J., van Bree C., et al. Radiation-enhanced vascular endothelial growth factor (VEGF) secretion in glioblastoma multiforme cell lines — a clue to radioresistance? J Neurooncol 2005; 74(2): 99–103, http://dx.doi.org/10.1007/s11060-004-4204-7.
- Ueda R., Iizuka Y., Yoshida K., et al. Identification of a human glioma antigen, SOX6, recognized by patients' sera. Oncogene 2004; 23(7): 1420–1427, http://dx.doi.org/10.1038/sj.onc.1207252.
- Ueda R., Ohkusu-Tsukada K., Fusaki N., et al. Identification of HLA-A2- and A24-restricted T-cell epitopes derived from SOX6 expressed in glioma stem cells for immunotherapy. Int J Cancer 2010; 126(4): 919–929, http://dx.doi.org/10.1002/ijc.24851.
- Pellegatta S., Poliani P.L., Corno D., et al. Neurospheres enriched in cancer stem-like cells are highly effective in eliciting a dendritic cell-mediated immune response against malignant gliomas. Cancer Res 2006; 66(21): 10247–10252, http://dx.doi.org/10.1158/0008-5472.CAN-06-2048.
- Xu Q., Liu G., Yuan X., et al. Antigen-specific T-cell response from dendritic cell vaccination using cancer stem-like cell-associated antigens. Stem Cells 2009; 27(8): 1734–1740, http://dx.doi.org/10.1002/stem.102.










