Novel Biocompatible Material Based on Solid-State Modified Chitosan for Laser Stereolithography
The aim of the investigation was to develop a novel biodegradable material based on chitosan synthesized by solid-state technoogy, and to create based on biocompatible three-dimensional cell-carrying scaffolds using laser stereolithography.
Materials and Methods. Reactive systems were developed based on chitosan grafted with allyl, polyethylene glycol diacrylate, and the photoinitiator Irgacure 2959. The structures were obtained using laser stereolithography setting LS-120 (Institute on Laser and Information Technologies, Russian Academy of Sciences, Russia).
Results. Partial replacement of chitosan amino groups by allyl groups (CТ-А) and the introduction of polyethylene glycol diacrylate (PEG-DA) as a crosslinking agent were found not to reduce the material biocompatibility. The metabolic activity determination of NCTC L929 cells using MTT assay showed that the samples under study to contain none water-soluble components toxic to mammalian cells. The samples based on CT-A and CT-A with a crosslinking agent PEG-DA are biocompatible and are able to support adhesion, spreading and proliferative activity of human mesenchymal stromal cells, but have significant differences in the extent and nature of the expression activation of gene markers for osteogenic differentiation path.
Recently, researches and developments in biomedical material science are growing rapidly aiming at solving the problems of regenerative medicine. According to WHO, their relevancy is due to the increase in the number of traumas and injuries, since their therapy is related to the use of substitution implants. The recovery of normal functioning of a living system by tissue growth initiation is known to be more effective than the techniques related to the application of substitution structures (prosthetic repair, etc.) [1]. Several methods are used to form three-dimensional structures containing bioactive components, among others: three-dimensional printing [2], selective laser sintering [3]. Laser stereolithography is a promising technique based on the initiation of local three-dimensional cross-links between reactive components of macromolecules when exposed to laser radiation in the ultraviolet spectrum. The technique enables to develop structures of required architectonics based on a computer model that can be developed using both specific computer support, and also the data obtained by in vivo methods of three-dimensional structure analysis of an object (e.g., data on MRT tissue defects when manufacturing an appropriate polymer implants) [4]. Three-dimensional matrix-carriers (scaffolds) based on gelatin [5], hyaluronic acid [6], fibrin [7] were fabricated using laser stereolithography. To apply this technique in regenerative medicine we need to develop a wide range of biocompatible photopolymeric printing compositions.
The aim of the investigation was to develop a novel biodegradable material based on chitosan synthesized by solid-state technology, and to create based on biocompatible three-dimensional cell-carrying matrices using laser stereolithography.
Materials and Methods
Solid-state chitosan modification. Solid-state synthesis of unsaturated simple ether of chitosan — allyl chitosan (CHT-A) — was carried out through the interaction of chitosan and allyl bromide (99%; Sigma-Aldrich, Germany) in the absence of any liquid dispersion media under the conditions of shear deformation in Berstorff ZE40 (Germany). The extruder was equipped with co-rotating processing elements providing compression and shift of the material in a thin layer. Chemical interaction of the components under forced plastic flow under such conditions results in the formation of high-yield products [8]. In accordance with the work [9], we used chitosan obtained through solid-state synthesis by alkaline deacetylation of crab shell chitin (VOSTOK-BOR, Russia). The content of residual acetamide (chitinious) components in the initial chitosan sample was 10%, the average degree of chitosan polymerization in a macromolecule being 360 (Мw=60 kDa). We used an alkylating agent, 0.5 mol, per a chitosan component and treated reaction mixtures at –5°С. The products were washed with n-isopropanol and dried in a vacuum oven at 50°С to remove completely unreacted monomer.
Photocurable composition preparation. For a photosensitive composition we prepared 5% CHT-А solution in 4% acetic acid. Insoluble fraction (unmodified chitin with high crystallinity) was separated by centrifugation (40 min, 14,500 rpm) followed by membrane filtration, pore size being 0.45 μm. Then we placed the solution, 120 ml, in an evaporating weighing bottle (35°С; 0.2 MPa) for an hour to remove a part of the solvent, and added polyethylene glycol diacrylate (PEG-DA, Sigma-Aldrich, Germany) 15 wt-% and mixed at room temperature for 30 min. The resulting hydrogel was added a photoinitiator Irgacure 2959, 1 wt-% (BASF Kaisten AG, Germany). The system was mixed at room temperature for 2 h till the homogeneous solution.
Preparation of samples for cell tests. The prepared composition was applied on the surface of cover glasses, 12 mm in diameter (the sample surface area was 1.13 cm2). Before applying hydrogel, cover glasses were washed with water and embedded in 5% HCl aqueous solution for 6 h followed by 20-minute distilled water wash in an ultrasound bath. UV induced cross-link of hydrogels was performed for 120 min exposed to UV mercury vapor lamp DRSh-100 (Russia). Two type materials were prepared: clear CHT-А and CHT-А with a cross-linking agent PEG-DA.
Cell tests. Before tests all samples were sterilized by 70% alcohol within 3 min followed by wash with a sterile culture medium DМЕМ/F-12 with penicillin, 100 U/ml and streptomycin, 100 μg/ml, added. Biocompatibility was studied in vitro using extractions and cell culture on the surface of the materials themselves.
Culture medium DМЕМ/F-12 with penicillin, 100 U/ml, and streptomycin, 100 μg/ml, added was used as a model medium to prepare extractions. Extractions were obtained in compliance with aceptics for 3 days at 37°С, each material being tested thrice. The ratio of material surface area and model medium volume was 1.13 cm2/1 ml of medium. Cytotoxic effect of soluble impurities contained in material extracts on NCTC L929 fibroblasts was assessed using МТТ assay according to the described procedure [10].
To determine adhesion characteristics of materials and their interaction with anchorage-dependent cells we used mesenchymal stem cells (MSC) isolated from abortive autopsy material of human fetuses according to the previously developed technique [11]. The cells were cultured in DMEM/F-12 (1:1, Life technologies, USA) also containing: 10% FBS (Fetal Bovine Serum, HyClone, USA), L-glutamine (2 mmol), penicillin (100 U/ml), streptomycin (100 μg/ml), vitamin solution (PanEco, Russia) at 37°С under the atmosphere of 5% СО2. When growing and reaching the subconfluent condition, the cells were treated with 0.25% trypsin-EDTA solution and subcultured (passage) in new bottles (cell mass of one bottle was divided among two bottles). For assay we used the cells on passage 4.
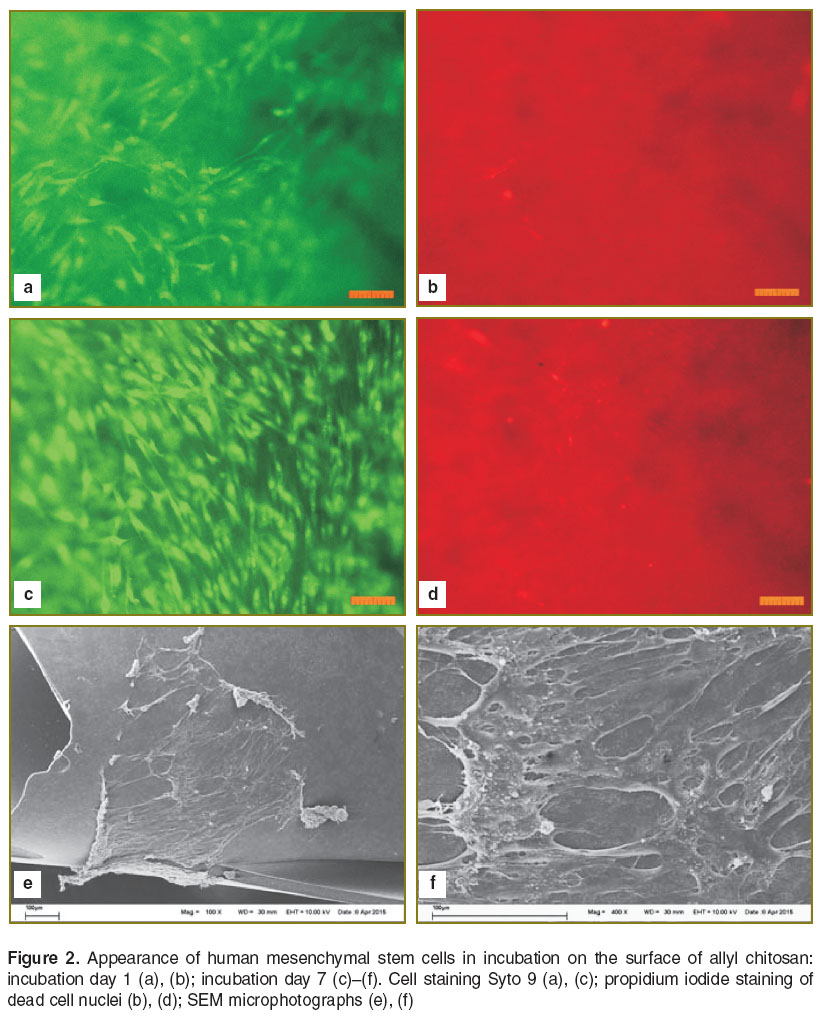
The cells were cultured on the surface of the samples under study, the density being 5·104/cm2, and cultured within 7 and 23 days. Their morphology and viability were estimated under the microscope Axiovert 200 (Karl Zeiss, Germany) using a staining set L-7007 LIVE/DEAD BacLight Bacterial Viability Kit (Invitrogen, USA) for fluorescent staining.
After culture we prepared the samples to study using scanning electron microscopy (SEM): the samples were washed in 0.1 М phosphate-buffered saline (PBS) (pH=7.4) and fixed in 2.5% buffered glutaric dialdehyde solution for 12 h at 5°С. After fixation the gel samples were washed with PBS and dehydrated at 4°С successively in battery of high proof ethyl alcohol 50, 75, 80, 90% and in absolute ethyl alcohol at a final stage. At each stage the samples were twice put in a corresponding alcohol solution for 5 min. To remove alcohol the samples were transferred into hexamethyldisilazane for 30 min and then air dried.
Real time polymerase chain reaction procedure. To isolate general messenger RNA from cells we used the kit Isolation of a full-size poly(А) mRNA on magnetic particles (Silex, Russia). The resultant mRNA was used to synthesize complementary DNA (cDNA) by means of the kit Synthesis of the first cDNA (oligo(dT)15) chain (Silex, Russia), while cDNA resulted from reverse transcription was used as a matrix for real time polymerase chain reaction (PCR).
PCR was carried out using ABI Prism 7500 (Applied Biosystems, USA) and an intercalating agent SybrGreen and a reference dye ROX. We studied the expression of 22 marker genes reflecting osteogenic differentiation (See the Table). Marker genes were selected from the database for PCR arrays Qiagen (Germany) to determine osteogenesis (http://www.sabiosciences.com, “human osteogenesis PCR array” PAHS-026Z), as well as the database Gene Ontology (ID: 000164) [12, 13]. Primers for each marker gene were selected using the program PrimerEpress (Applied Biosystems, USA).
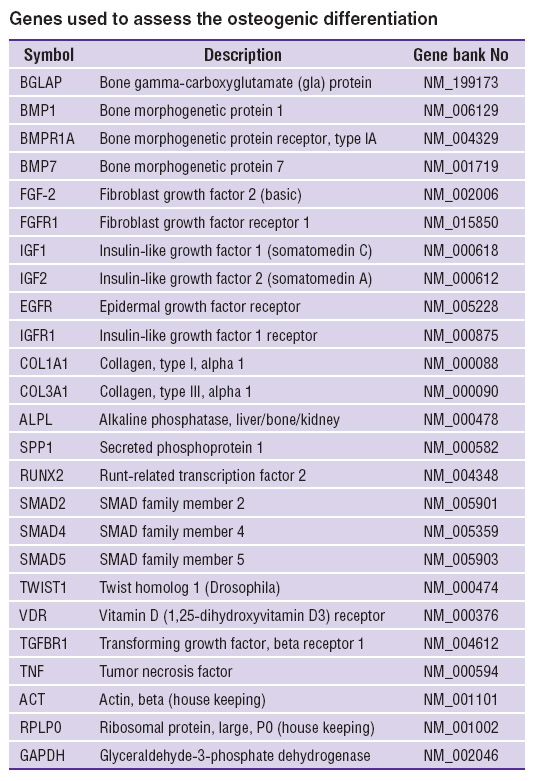 Genes used to assess the osteogenic differentiation Genes used to assess the osteogenic differentiation
|
Concentration of primers in optimization reactions was 0.05 pmol/μl, Mg2+ ion concentration ranged from 1.5 mmol, the enzyme concentration being 0.2 units per 20 μl. The length of primers averaged 24 nucleotides. The annealing temperature was 60°С, the length of an amplifying fragment was 94–100 base pairs. To check the reaction specificity we put the amplification products to 2% agarose electrophoresis test and used amplicon temperature dissociation curves.
Real-time PCR findings were analyzed by threshold fluorescence using ΔΔС(Т) technique and a Web-site to study data PCR Arrays QIAGEN (http://www.sabiosciences.com).
As a reference we used the gene expression of actin genes (ACT), a large ribosomal protein subunit (RPLP0) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
Data processing. The findings of cell tests and swelling were statistically processed using the program Origin. Root-mean-square deviation from mean value was taken for error, the differences according to Mann–Whitney U-test were taken for significant, if p<0.05.
Laser stereolithography. All samples were prepared on an experimental model of laser stereolithography apparatus LS-120 (Institute on Laser and Information Technologies, Russian Academy of Sciences, Russia). Layer thickness in growing samples was 200 μm. Structurization was performed using a HeCd-laser (wavelength was 325 nm, radiation power was 15 mW). We determined the layer formation rate based on the laser power and the technological parameters of the composition curing deduced from experiments: Ec=50 mJ/cm2 (a parameter characterizing a threshold value of exposure dose for solid polymer film formation start) and Dp=0.15 mm (a parameter to characterize critical thickness of a film).
Swelling characterization. We studied the swelling characteristics of three-dimensional structures according to the developed algorithm. According to the above described procedure we placed three-dimensional structures (without swabbing) in ethyl alcohol (rectificate) or 25% ammonia for 2 h. Then the samples were put into distilled water or PBS for 9 days, and during this period the scaffolds swelled to the maximum water content. To model the behavior of structures in vivo we changed a solvent every 48 h during the swelling process. Before weighing the structures were placed on nonwoven fabric for 10 min to remove excess water. The procedure was repeated for three identical samples to the constant weight value. Swelling rate (SWR) was determined according to the formula:
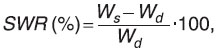
where Wd is matrix weight after spray-freeze drying; Ws is weight of swollen matrix.
Swelling was also measured in PBS and distilled water without any solvents.
Results and Discussion
Synthesis of reactive polymeric systems based on chitosan. Chitosan is a cationic polysaccharide having primary amine group almost in each polymeric unit. This determines its biological activity as well as makes it the most attractive polymer among many polysaccharides to perform their chemical modification. To increase the reactivity of chitosan functional groups under laser-induced cross-linking, unsaturated groups were introduced into chitosan structure via its interaction with allyl bromide under the conditions of shear deformation in an extruder. We chose solid-state synthesis which makes it possible to conduct chemical modification of polymers in the absence of any liquid dispersion media as well as any catalysts and initiators of the processes, since it is the most acceptable technique to solve the problem of thermodynamic incompatibility of hydrophilic chitosan and hydrophobic modifier. Chitosan amino groups are much more reactive than hydroxyl ones in the course of non-catalyzed nucleophilic replacement reactions, while alkaline catalyze results in the formation of mixed O- and N-substituted allyl derivatives [14, 15]. As it follows from NMR (nuclear magnetic resonance) and FTIR (Fourier transform infrared) findings, the substitution of chitosan amino groups occurs mainly under the given conditions of synthesis in accordance with the scheme given bellow:
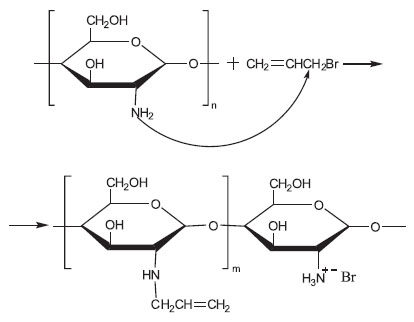
The analysis of the 1H NMR spectrum of an allyl chitosan sample in D2O solution doped with 1% HCl (recorded on a Bruker Avance II 300 spectrometer, Germany) showed the content of allyl-substituted units to be 8–10%. Chemical shift of carbon in allyl methylene group (at 53 ppm) in 13C–{1H} APT (attached proton test) spectrum (75.5 MHz) indicated its binding with nitrogen atom and showed preferential N-alcylation of the substrate. At the same time, chitosan modified by hydrophobic unsaturated substituents retained solubility in acidic aqueous media characteristic of the initial polymer.
Electronic absorption spectra of 1% polymer solution in 0.1 М HCl were recorded by a Shimadzu UV 2501 PC spectrophotometer (Japan) using quartz cuvettes with 10 mm optical path length. The spectral bands referring to absorption and Rayleigh scattering were mathematically separated followed by absorption spectra decomposition into separation bands. The absorption spectra of allyl chitosan were in the range of 200–420 nm and consisted of three intense bands with maximum at 264, 301 and 351 nm; their intensity ratio being 0.3/1/0.1 (while the initial chitosan spectrum showed the bands with maximum at 252, 299 and 346 nm and the intensity ratio of 1/1/0.4). Thus, in the allylation product spectrum a constituent with maximum 301 nm prevailed, its intensity being twofold compared to the initial chitosan, while the intensity of two other bands was significantly decreased. Since the electron absorption spectra of the diluted solutions of chitosan and allyl chitosan showed no significant difference, we assume the absence of aggregation effect in both polymeric solutions in the concentration range from 0.125 to 1%.
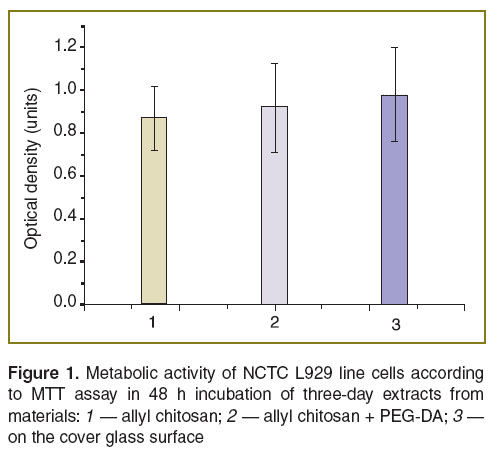
Biocompatibility and biological activity of the chitosan-based composition. We studied metabolic activity of NCTC L929 line cells in the presence of extracts from CHT-А and CHT-А with PEG-DA using МТТ assay based on the capability of mitochondrial dehydrogenases to convert water-soluble 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl-2Н-tetrazolium bromide into formazan, its quantity being determined photometrically by optical density of the solution when exposed to light source with wavelength of 540 nm. The study showed no significant differences between the controls and 3-day extracts from samples indicating no water-soluble toxic components in the engineered compositions (Figure 1).
Human MSC were used to determine adhesion characteristics of the materials and their interaction with substrate dependent cells. The analysis of morphological traits and viability of human MSC cultured on the surface of CHT-А and CHT-А with PEG-DA demonstrated the cell death percentage not to exceed 1–2%. Cells spread and proliferated on the surface of both materials under study. The morphology of cells was no different from control, though the density of a cell monolayer on a cover glass on day 7 was significantly higher compared to that on polymer films (Figures 2–4). To assess the effect of physicochemical characteristics of the materials on differentiating activity of human MSC we determined a phenotypic cell profile at different culture stages. Since the analysis of 22 major genetic markers (See the Table) [12, 13] is generally used to reflect the process of osteogenic differentiation, the present study involved real-time PCR to analyze their expression.
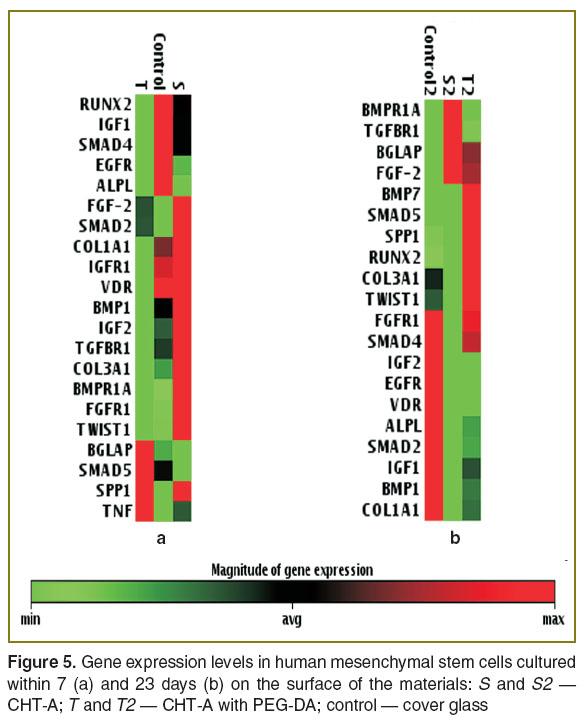
The study of the cell cultured on the glass and on the polymers under study showed the differences in expression activation degree of genetic markers of osteogenic differentiation on day 7 (Figure 5 (а)). In controls (culture on the glass) and on allyl chitosan there was expression activation and a high mRNA level of genes ALPL, FGFR1, TWIST, BMP1, IGFR1, VDR and TGFBR1. However, the cells cultured on CHT-А with PEG-DA expressed the genes under study poorly, their BGLAP and SMAD 5 levels being high. At this stage, the cells cultured on all the materials were found to have no BMP7 transcription.
A longer cell culture (23 days) on the test materials slightly changed the gene transcription (Figure 5 (b)). The cells cultured on all the materials had no TNF expression. On CHT-A samples only a few genes had a high expression level compared to the cells cultured on the cover glass. A control group, in general, showed the same transcription pattern of marker genes, though the majority of genes enhanced it significantly. In addition, the number of mRNA genes IGFR1, VDR and TGFBR1 decreased. Cell culture on CHT-А with PEG-DA samples stimulated the transcription of genes with somewhat different pattern compared to that in the controls. These cells increased the expression level of BGLAP, FGF-2, SPP1, SMAD5, COL3A, TWIST1; and BMP7 appeared, while the transcription of genes IGF2, EGFR and VDR was low.
Thus, as early as the initial cell culture stages allyl chitosan induced osteogenic differentiation genes, while CHT-А with PEG-DA exhibited this property only in a prolonged cell culture.
Matrices produced by laser stereolithography. CHT-A based matrices produced by laser stereolithography are structurally uniform material, the samples are in the form of crossed helixes (Figure 6) or two superimposed circles with centered beams, and a hole (wheel-shaped). Under mild exposure original matrices recover their former shape. When drying out, the strength of samples increases, though their elasticity decreasing and fragility growing.
 Figure 6. Appearance of structured matrices; 1 scale mark = 1 mm Figure 6. Appearance of structured matrices; 1 scale mark = 1 mm
|
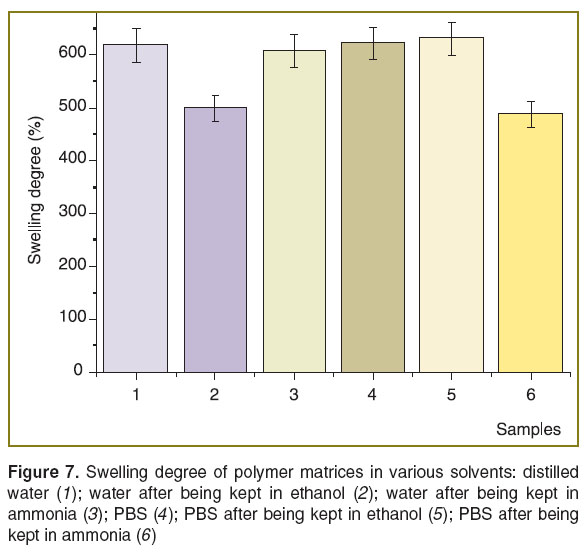
A method to control swelling rate of developed three-dimensional structures. An important task when generating the materials for biomedical three-dimensional structures is to develop compositions to control the swelling rate of matrices based on these structures when used in vivo [16]. Substitution therapy when such structures are introduced in damaged tissues when performing surgery can both stimulate healing and also reduce edema removing excessive bioliquid from trauma [17]. For the materials and structures on their base we developed an approach to control swelling rate of scaffolds in the range from 490 to 630 wt-%.
The algorithm of swelling analysis is described in the section “Materials and Methods”. High swelling rate (up to 630%) showed the matrices preliminary kept in ammonia and then put into distilled water. Mean swelling rate was 630% when PBS was used as a solvent for samples kept for 2 h in ethanol. Statistical processing of the findings showed that empiric values (Uemp) of these samples are insignificant (Figure 7, columns 1, 3, 4, respectively).
A sample was preprocessed by ammonia to reduce the swelling rate in a buffer, chitosan developing into a basic form. The absence of an acetate counter ion on an amino group reduces the sorption capacity of matrices relating to liquid, and by that allowing removing excessive swelling.
Similar SWR value was obtained for matrices swelled in distilled water and initially kept in ethyl alcohol (Figure 7, columns 2 and 5). Ethanol also slightly compresses the structures compared to those treated with ammonia and nontreated. It occurs due to the displacement of bound water molecules from a polymer by ethanol molecules. Figure 8 shows modified chitosan-based three-dimensional structures including an original sample, as well as the samples after freeze drying and preliminary kept in PBS and NH4OH.
Conclusion. We developed reactive chitosan derivatives by the interaction of chirosan and allyl bromide under shear deformation in an extruder without solvents. A number of films were formed based on the obtained compounds resulted from UV-induced cross-linking in the presence of a photoinitiator and a cross-linking agent. The study of metabolic activity of NCTC L929 cells using MTT assay showed the samples under study to contain none water-soluble components toxic to mammalian cells. Novel materials — allyl chitosan and allyl chitosan with polyethylene glycol diacrylate — are biocompatible and able to equally provide adhesion, spreading and proliferative activity of human mesenchymal stem cells, though have significant differences in the extent and nature of the expression activation of gene markers for osteogenic differentiation path. On the basis of the developed material by laser stereolithography we fabricated three-dimensional matrices with controlled swelling rate in phosphate-buffered saline.
Study Funding. The study was supported by Russian Research Fund, project No.15-13-00140 (as related to laser stereolithography and the study of scaffold cytotoxicity) and Russian Foundation for Basic Research, a project No.14-29-07234-офи_м (as related to modified chitosan synthesis).
Conflicts of Interest. The authors have no conflicts of interest related to the present study.
References
- Regenerative Medicine. Editor by Steinhoff G. Springer Science+Business Media B.V.; 2011; 1032 p., http://dx.doi.org/10.1007/978-90-481-9075-1.
- Lam C.X.F., Moa X.M., Teoh S.H., Hutmacher D.W. Scaffold development using 3D printing with a starch-based polymer. Materials Science and Engineering: C 2002 May; 20(1–2): 49–56, http://dx.doi.org/10.1016/S0928-4931(02)00012-7.
- Chumnanklang R., Panyathanmaporn T., Sitthiseripratip K., Suwanprateeb J. 3D printing of hydroxyapatite: effect of binder concentration in pre-coated particle on part strength. Materials Science and Engineering: C 2007 May; 27(4): 914–921, http://dx.doi.org/10.1016/j.msec.2006.11.004.
- Mankovich N.J., Samson D., Pratt W., Lew D., Beumer J. Surgical planning using three-dimensional imaging and computer modeling. Otolaryng Clin North Am 1994 Oct; 27(5): 875–889.
- Koroleva A., Kufelt O., Schlie-Wolter S., Hinze U., Chichkov B. Laser microstructured biodegradable scaffolds. Biomedizinische Technik/Biomedical Engineering 2013 Oct; 58(5): 399–405, http://dx.doi.org/10.1515/bmt-2013-0036.
- Leach J.B., Bivens K.A., Collins C.N., Schmidt C.E. Development of photocrosslinkable hyaluronic acid polyethylene glycol-peptide composite hydrogels for soft tissue engineering. J Biomed Mater Res A 2004 Jul; 70A(1): 74–82, http://dx.doi.org/10.1002/jbm.a.30063.
- Koroleva A., Gittard S., Schlie S., Deiwick A., Jockenhoevel S., Chichkov B. Fabrication of fibrin scaffolds with controlled microscale architecture by a two-photon polymerization–micromolding technique. Biofabrication 2012 Mar; 4(1): 015001, http://dx.doi.org/10.1088/1758-5082/4/1/015001.
- Prut E.V., Zelenetskii A.N. Chemical modification and blending of polymers in an extruder reactor. Russian Chemical Reviews 2001; 70(1): 65–79, http://dx.doi.org/10.1070/RC2001v070n01ABEH000624.
- Akopova T.A., Rogovina S.Z., Vikhoreva G.A., Zelenetskiy S.N., Gal'braykh L.S., Enikolopov N.S. Formation of chitosan from chitin under shearing strain. Vysokomolekulyarnye soedineniya. Seriya B 1991; 32(10): 735–737.
- Filippov Ya.Yu., Larionov D.S., Putlyaev V.I., Sokolov A.V., Koval’kov V.K., Agakhi K.A., Selezneva I.I., Nikonova Yu.A. Reaction-associated resorbable phosphate materials: production and testing in vitro. Glass & Ceramics 2013 Nov; 70(7–8): 306–310, http://dx.doi.org/10.1007/s10717-013-9568-8.
- Selezneva I.I., Savintseva I.V., Vikhlyantseva E.F., Davydova G.A., Gavrilyuk B.K. Immobilization and long-term culture of murine embryonic stem cell in collagen-chitosan gel matrix. Kletochnye tekhnologii v biologii i meditsine 2006; 3: 135–140.
- Twine N.A., Chen L., Pang C.N., Wilkins M.R., Kassem M. Identification of differentiation-stage specific markers that define the ex vivo osteoblastic phenotype. Bone 2014 Oct; 67: 23–32, http://dx.doi.org/10.1016/j.bone.2014.06.027.
- Ho N.C., Jia L., Driscoll C.C., Gutter E.M., Francomano C.A. A skeletal gene database. J Bone Miner Res 2000 Nov; 15(11): 2095–2122, http://dx.doi.org/10.1359/jbmr.2000.15.11.2095.
- Nud’ga L.A., Petrova V.A., Denisov V.M., Petropavlovskiy G.A. Chitosan alkylation. Zhurnal prikladnoy khimii 1991; 64(1): 229–232.
- Xia Y., Guo T., Zhao H., Song M., Zhang B., Zhang B. A novel solid phase for selective separation of flavonoid compounds. J Sep Sci 2007 Jun; 30(9): 1300–1306, http://dx.doi.org/10.1002/jssc.200600376.
- Kerker J.T., Leo A.J., Sgaglione N.A. Cartilage repair: synthetics and scaffolds: basic science, surgical techniques, and clinical outcomes. Sports Med Arthrosc 2008; 16(4): 208–216, http://dx.doi.org/10.1097/JSA.0b013e31818cdbaa.
- Tollar M., štol M., Kliment K. Surgical suture materials coated with a layer of hydrophilic Hydron gel. J Biomed Mater Res 1969 Jun; 3(2): 305–313, http://dx.doi.org/10.1002/jbm.820030210.