Brain–Computer Interfaces with Intracortical Implants for Motor and Communication Functions Compensation: Review of Recent Developments
Brain–computer interfaces allow the exchange of data between the brain and an external device, bypassing the muscular system. Clinical studies of invasive brain–computer interface technologies have been conducted for over 20 years. During this time, there has been a continuous improvement of approaches to neuronal signal processing in order to improve the quality of control of external devices. Currently, brain–computer interfaces with intracortical implants allow completely paralyzed patients to control robotic limbs for self-service, use a computer or a tablet, type text, and reproduce speech at an optimal speed. Studies of invasive brain–computer interfaces regularly provide new fundamental data on functioning of the central nervous system. In recent years, breakthrough discoveries and achievements have been annually made in this sphere.
This review analyzes the results of clinical experiments of brain–computer interfaces with intracortical implants, provides information on the stages of this technology development, its main discoveries and achievements.
Introduction
Brain–computer interfaces (BCIs, neural interfaces) allow direct information exchange between the brain and the computer with the data transfer to an external technical device. Such interfaces include electrodes to record brain activity signals, a signal processing system (filtering, feature extraction, decoding, classification and conversion into a control command), as well as a controlled external technical device [1]. In case of invasive BCIs, the system can also send a signal in the opposite direction: from external sensors to neural implants in the cerebral cortex, thus ensuring neuromodulation (see the Figure) [2].
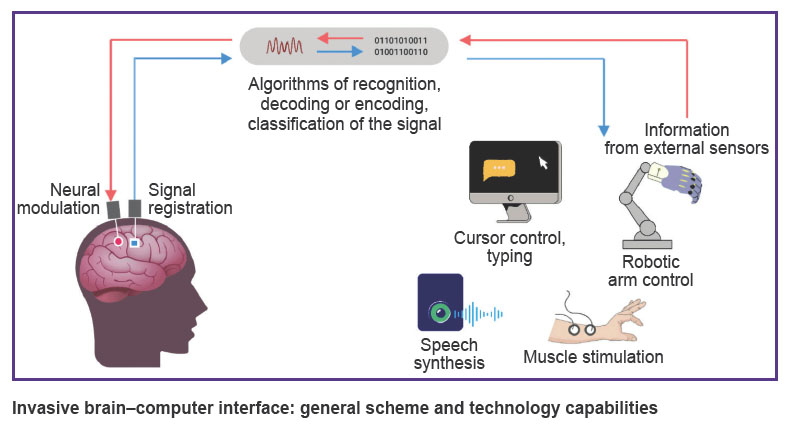
|
Invasive brain–computer interface: general scheme and technology capabilities |
In recent years, an extensive evidence base on the use of non-invasive BCIs in rehabilitation after stroke has been formed [3–13]. Signal recording in such interfaces is conducted from the head surface, most often using electroencephalographic (EEG) sensors or near-infrared spectroscopy (NIRS) during motor imagery practice [14, 15]. Non-invasive BCIs allow motor imagery practice to stimulate neuroplasticity and restore motor function provided that the patient has rehabilitation potential [10–13].
The signals recorded in invasive BCIs are the local field potential (using extracortical or intracortical sensors) and neurons spiking activity (using intracortical sensors) [16]. Despite high cost of development and research, as well as the need for surgical intervention, invasive BCIs are no-compromise means of interaction with the environment for completely paralyzed and speechless patients, who preserve cognitive functions in case of tetraplegia and anarthria or the locked-in syndrome of various etiologies. Unlike non-invasive BCIs, the evidence base for which includes multiple randomized clinical trials and their meta-analyses, the clinical application of invasive neural interfaces is limited to several dozen cases only. However, almost each new case is a scientific breakthrough, and the corresponding articles are published in the top-rated journals [17–27].
Currently, low or minimally invasive BCIs with extracortical [17, 26, 28, 29] or endovascular [30, 31] sensors are being developed; but advances in development of neural interfaces with intracortical sensors [2, 32] are of greater scientific interest. The obvious advantages of such BCIs include the following: 1) getting signals of brain activity with the highest temporal and spatial resolution; 2) high signal-to-noise ratio; 3) the closest or the most precise placement of electrodes in the target brain areas; 4) transmission of the signal in the opposite direction — from external sensors to the cerebral cortex [2, 32]. Due to high, one-neuron only, spatial resolution of signal recording or neuromodulation, intracortical neural implants allow to obtain new data on localization of specific functions in the cerebral cortex and the characteristics of its neurons functioning [16, 24, 25, 33, 34].
The aim of this review is to analyze and describe the capacities of brain–computer interfaces with intracortical implants in rehabilitation of patients with severe motor disorders.
Literature search methodology
Literature search was conducted in the MEDLINE (PubMed) database using the search query: ((invasive[tiab] OR intracortical[tiab]) AND (brain-computer[tiab] OR brain-machine[tiab] OR “neural interface*”[tiab]) OR intracortical implant*[tiab]) AND humans[mh]. Additionally, a literature search was conducted in the eLIBRARY.RU system using the keywords: “brain–computer interface”, “neural computer interface”, “neural interface”. The date of search was July 15, 2023.
Articles were selected for analysis based on the following criteria: 1) articles or letters to the editor published in the peer-reviewed scientific journals; 2) publications on the use of invasive BCIs with intracortical sensors in humans; 3) articles on using BCIs to compensate for motor or speech dysfunctions.
Preclinical studies and first experiments
Studies on the use of implanted sensors to record signals from the monkey cerebral cortex started in the 1960s [35, 36]. In the 1970s, in experiments with monkeys, one managed to create a system for converting cortical signals into cursor movement in real time [37, 38]. The animals were able to control the cursor by modulating signals from the motor cortex, even without an actual movement. In the late 1990s and early 2000s, researchers of preclinical studies applied BCI systems with robotic limbs as an externally controlled device [39–47]. With these technologies, animals could feed themselves. After the start of clinical experiments of invasive BCIs, animal studies continue to test several scientific hypotheses and search for new approaches to signal processing [48–57].
It is believed that the first experiment related to control of an external device by signals from a neural implant in the human brain was conducted in 1963 by British neurosurgeon Grey Walter [58, 59]. The researcher wanted to test the hypothesis that the intention to perform an action is accompanied by certain bursts in neuronal activity. Patients, who previously had electrodes implanted in the motor cortex of their brain for medical reasons, were asked to switch projector slides by pressing a button. Here, the button was dummy, though the patients were not aware of that. In fact, the slides were switched by an amplified signal from the neuroimplant. The patients were surprised that the slide projector anticipated their actions.
First clinical studies, neurotrophic electrode
In the late 1990s, researchers led by P.R. Kennedy were the first to implant neurotrophic electrodes into several patients with tetraplegia for long-term recording of cortical signals [60–62]. The electrodes consisted of two insulated gold wires inside a glass cone of 1.5 mm long and 0.1–0.4 mm in diameter having autologous neurotrophic factors. This was a wireless system [63]. 1.5–3 months after implantation, the neuron processes in the cerebral cortex grew into the tip of the electrode with subsequent myelination. A few weeks after implantation, the first signals could be recorded, and within 1.5–3 months the signal became stable. Using neurotrophic electrodes, one managed to record a signal that remained stable for at least four years [64].
The first implantation of such a neurotrophic electrode was performed in a woman at the late stage of amyotrophic lateral sclerosis (ALS) [60]. Localization of the implantation site, the hand representation area in the right motor cortex, was determined using the functional magnetic resonance imaging (fMRI) during the imagination of hand movements and speech articulation. Almost immediately after the neuronal activity signal stabilization, the patient learned to control the cursor in the vertical direction. The implant had been functioning for 76 days — until the last days of the patient’s life. Then, the neurotrophic electrodes were implanted into other patients with tetraplegia and anarthria: in 1998, into a 53-year-old man 3 months after he had a brainstem stroke [61, 62], in 1999, into a 40-year-old man with a 12-year history of progressive mitochondrial myopathy [62], and in 2004, into a 26-year-old man who had a 5-year brainstem stroke [64, 65]. Using the invasive interface, the first of these patients managed to control the cursor in various directions on the display, could click the target and control the fingers of a virtual hand. The most interesting observation for the researchers was the ability to control the cursor without the need to imagine movements or any other standard brain paradigms: the patient controlled the cursor by self-will. Researchers associated this phenomenon with neuroplasticity, and called the electrode implantation area the “cursor cortex” [61]. However, the patient needed six months to master the typing skill after stabilization of the recorded signal, and the typing speed was three characters per minute. At that time, it was already possible to type text with the same speed using non-invasive BCIs.
During the experiment, the patient with mitochondrial myopathy developed a severe cognitive impairment due to disease progression, but was able to control the cursor in one direction [62].
In 2004, in order to interpret speech-associated neural activity, a neurotrophic electrode was implanted into a 26-year-old patient with brainstem stroke [64, 65]. The implant was grafted into a region of the cortex involved in planning the sound articulation. In this BCI with a Kalman filter decoder, neural signals generated during speech attempts were used to control a speech synthesizer. The accuracy of vowel sound reproduction by the patient after 25 training sessions was 70%.
Thus, the studies led by P.R. Kennedy developed a methodology to identify a cortical area for sensor implantation in patients with plegia and anarthria. For the first time, recording electrodes were implanted into the human cerebral cortex for a long period of time, as well as the researchers demonstrated: the electrodes safety and feasibility for using to control a cursor, a hand avatar, and a speech synthesizer through voluntary modulation of cortical signals even years after the plegia onset. Despite the limited functionality of the first invasive BCIs, in the late 1990s the possibility of creating alternative means of communication and self-care for patients with tetraplegia was shown [60–62, 64, 65].
Early research under the BrainGate project
In the early 2000s, the BrainGate project launched a series of clinical studies of invasive BCIs. The neuroimplant was an array of 100 silicon microelectrodes (96 active), 1.5 mm long, arranged in a 10×10 pattern on a 4×4 mm platform (Blackrock Microsystems, Salt Lake City, Utah, USA) — the so-called “Utah array” [66]. Before that, the microimplant had been studied in preclinical studies [67–70].
The first patients to whom BrainGate sensors were implanted in 2004–2005 were 25 and 55 year old men with spinal cord injury (SCI) at the level of the fourth cervical vertebra (C4 ASIA A in line with the American Spinal Injury Association scale), which had been occurred 3 and 5 years before implantation, respectively [18, 71]. Researchers led by J.P. Donoghue demonstrated the capacity of this sensor to record both neurons spiking activity and local field potentials during 6.5 and 11 months (for the first and second patient, respectively), as well as the patients’ ability to control these signals years after the corticospinal tract breakage. Neural decoding in the BCI circuit allowed to open e-mail and control the TV using the “neural cursor”. The first patient successfully reached 73–95% of targets with the “neural cursor”; the average time to reach the target was 2.5 s [18, 71].
In further studies involving patients with brainstem stroke or ALS, signal processing algorithms were optimized, which allowed to reduce calibration time and to get a better cursor control [72–74].
Later, it was shown that even 1000 days (2.7 years) after implantation, the microelectrode array continued recording the signals: the patient demonstrated consistently high levels of control quality over five consecutive days of the experiment. The rate of successful target achievement in this experiment was 94.9% for the radial and 91.9% for the random target location (which exactly simulates the computer mouse). Of the 564 tasks, only 37 failed by timeout, but not due to cursor navigation errors [75]. These results took down the investigators’ concerns about the risk of fast decline in the electrodes function due to tissue reaction to the implant. By now, the even longer duration of functioning have been shown for neural implants [19].
Robotic arm control
Subsequent studies were mainly related to control of an external robotic multi-joint arm to do functionally significant movements (Table 1). This type of task involved manipulating an object in three dimensions along curved trajectory and sequential actions at different joints. Compared to cursor control, one needs more precise control of speed and movement pattern, as well as exact positioning and command planning.

|
Table 1. Main results of clinical studies of neural interfaces with a robotic arm |
In the experiment of 2012, two patients with tetraplegia controlled a multi-joint robotic arm and managed to reach out and grasp the target object in 47 and 62% of attempts [19]. By controlling a robotic arm, the experiment participant was able to bring a glass of coffee to her mouth and drink it with a straw. Owing to the BCI, she had performed a self-care act for the first time in 15 years. Despite the 5-year implantation period, the electrode array recorded signals sufficient to control the robot, but a decrease in their amplitude and a drop-down in active electrode channels were observed.
In another study [20], a patient with spinocerebellar ataxia had two sensors implanted in the cortical area representing finger and forearm muscles of the dominant hemisphere. The anthropomorphic robotic arm design allowed movements with seven degrees of freedom. Already on the second day of training, the patient was able to freely operate the robotic arm in three dimensions. Over the next 13 weeks, the control quality consistently improved. The movements were smooth and coordinated, at a speed close to the same of a healthy person’s arm. The robot control level allowed to perform subtle adjustments and manipulations with various balls, cubes, and sticks. On average, the targets were successfully achieved in 92% of attempts, and in the ARAT test (Action Research Arm Test — a test to assess arm movements in central paresis) 17 out of 27 scores were received with the robotic arm.
In subsequent clinical experiments, researchers improved the BCI system architecture and signal processing algorithms, which allowed to control a robotic arm with 10 degrees of freedom [76], improve the grasp quality [77, 79], and reduce the system calibration time from 10 to 3 min [80]. Moreover, additional cortical areas for electrode implantation were identified as sources of signals that were associated with movement planning [25].
In one of recent studies, the BCI design included two robotic arms to perform complex bimanual tasks [78]. To control this system, a 30-years-aged patient with SCI was implanted with six electrode arrays in both brain hemispheres. A semi-automatic system was used to self-feed manipulating a fork and knife: some specific movements were programmed, and some were controlled by brain signals. The patient successfully completed 85% of bimanual tasks.
Brain–computer interface with electrical stimulation of paralyzed muscles
Electrical stimulation of patient’s muscles by the modulation of motor cortical signals ensures a more natural reaction of the motor system [81]. By now, several studies of invasive BCIs with functional electrical stimulation (FES) of muscles were conducted.
First, researchers demonstrated the ability of a patient with long-term tetraplegia to control certain movements of a virtual arm by using a BCI and simulating the work of specific muscles. For motor simulation, the estimated parameters of muscle contractile force and arm weight were considered [82].
In subsequent FES studies, the researchers succeeded in achieving BCI-controlled functional arm movements [83, 27]. In the first of them, a participant with C5–C6 SCI and over four year tetraplegia managed to get control of six different movements of his own wrist and hand, and was also able to take a bottle, pour its contents into a glass, put the bottle down, and mix the contents of the glass with a stirrer on average in 42 s [83]. In this study, machine learning algorithms were used to process neuronal signals. Electrical stimulation of the paralyzed muscles was conducted using 130 electrodes in a flexible sleeve, which was wrapped around the right forearm. The training continued for 15 months (up to three training sessions per week). The average control accuracy was 70%. Clinical assessment showed that with BCI-FES the patient’s motor abilities corresponded to the level of spinal cord lesion of the C7–T1 level, which is two vertebrae below the actual damage. This improvement is significant in reducing the burden of care for patients with C5–C6 SCI, as the majority of them require assistance with daily activities, whereas patients with C7–T1 SCI can live more independently.
In the next study, a patient with consequences of high SCI (C4 ASIA A) was able to successfully drink a cup of coffee and self-feed using BCI–FES [27]. The training was conducted for 18 weeks with an average of 8 h per week. The FES system included 36 transcutaneous electrodes to stimulate the muscles of the hand, forearm, and shoulder. The “drink coffee” task required a series of sequential actions: 1) straighten the elbow; 2) relax the grip; 3) take a cup; 4) bend the elbow to bring the cup to the mouth; 5) take a sip using a straw; 6) straighten the elbow to return the cup; 7) loosen the grip. These processes in total took from 20 to 40 s, and 11 out of 12 attempts were successful.
Bidirectional brain–computer–brain interface
Bidirectional brain–computer–brain interface allows not only to record signals from the cerebral cortex, but also to modulate its activity. Such an interface additionally includes external tactile sensors and electrodes implanted in the somatosensory cortex. Sensory feedback is of key importance to the majority of motor tasks, providing information about the location of the limb, about touching an object, and characteristics of that object. Sensory and motor functions are not independent: the brain creates complex motor plans and compares the expected result with the sensory feedback in order to appropriately adjust the movement [84].
The advantages of bidirectional BCIs were demonstrated in [23]. A patient with C5–C6 SCI had two electrode arrays implanted into each of the motor and somatosensory cortex of the dominant hemisphere, and the touch and pressure sensors were built into the hand of the robotic arm in the BCI circuit. The patient first learned to control the robotic arm with the visual feedback only. Then, with the tactile feedback added, the quality and speed of the robotic hand movements improved within the first four sessions. This was quantitatively assessed using the ARAT test, the average score for which increased from 17 to 21, and the test completion rate decreased by over 2 times [23].
High-performance communication neural interfaces
Due to continuous developments in the accuracy, speed, and stability of neural cursor control, patients with implanted cortical BCI sensors can type text with a speed sufficient for communication. However, recent advances in neural communication interfaces allow to type text or reproduce speech without using a virtual keyboard. Over the history of communication neural interfaces with intracortical sensors development (in 2000–2023), the speed of text reproduction increased from 3 characters per minute [61], which is comparable to the EEG–BCI performance, to 60 words per minute [24], which is approximately the speech rate of a healthy person (Table 2).
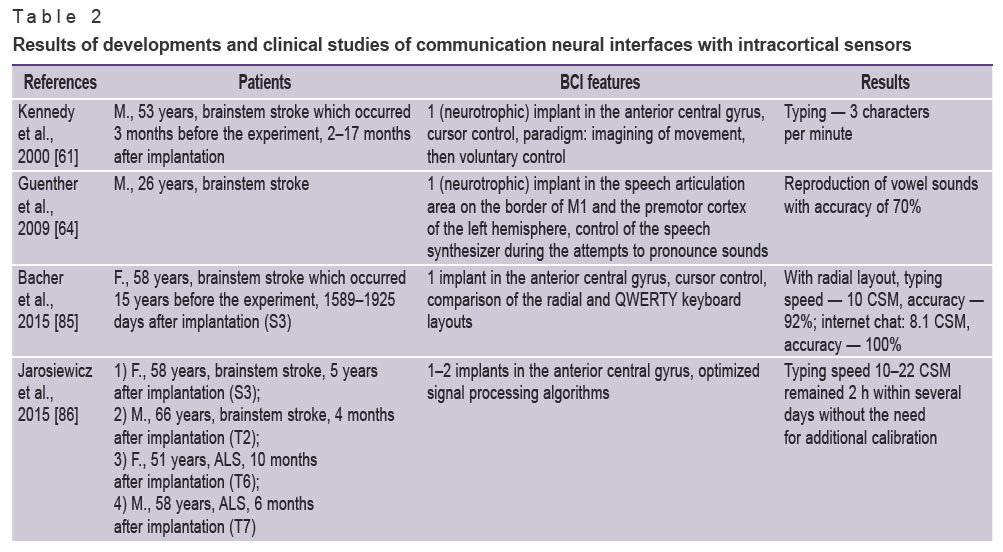
|
Table 2. Results of developments and clinical studies of communication neural interfaces with intracortical sensors |
At first, the keyboard layout was optimized to improve the performance of communication BCIs for the neural cursor control. The radial layout allowed to repeat text in the copy task at an average speed of 10 correct characters per minute and an accuracy of over 90% [85]. Keyboard layout optimization increased typing accuracy by 37–65%. These indicators were achieved by a patient with a 14-year history of anarthria and tetraplegia during three training sessions and a sensor implanted about 5 years before the experiment [85].
In further studies, optimization of approaches to signal processing allowed to achieve more stable control quality without the need for regular calibration [86] and increase the average typing speed to 39 correct characters (8 words) per minute; the maximum speed was 40.5 correct characters (9 words) per minute without auto-correction [79, 87]. The achieved typing speed was inferior to the communication performance of a healthy person in normal conditions: the typing speed on a smartphone is 115 characters (12–19 words) per minute, and the speech rate is 90–170 words per minute. However, the achieved speed of typing and cursor control allowed several paralyzed patients to use a tablet with a standard user interface to communicate in emails and chats, make search queries and use basic applications [88].
Further performance improvement of communication BCIs was achieved by applying a totally different paradigm for signal control. A patient paralyzed due to SCI imagined that he was writing words with a pen. The BCI system successfully learnt to recognize each letter, and the typing speed reached 90 characters (18 words) per minute with an accuracy of 94% in real time or >99% with autocorrection [22]. The paradigm of imagining the text-writing procedure turned out to be essentially simpler for the signal decoding than controlling the cursor movement to select letters. Researchers [22] believe that this is due to the fact that handwritten letters are easier to be distinguished than point-to-point movements as the spatiotemporal patterns of the letters neural activity vary more than straight line movements.
Application of another paradigm option — imagining typing with fingers on a virtual keyboard with a special symbol layout — allowed to achieve control accuracy of 95%. However, in terms of typing speed (14 characters per minute), this approach was inferior to typing by the neural cursor movement or using the imagining writing letters with a pen [90].
The latest publication to date, which is dedicated to neural communication interfaces with intracortical sensors, reports the development of a high-performance speech neural prosthesis [24]. A patient with ALS, who was unable to intelligibly speak, had 2 microelectrode arrays implanted in the Broca’s area and 2 in the ventral premotor cortex of the dominant hemisphere. The patient’s attempt to speak was interpreted by the BCI system at a speech rate of 62 words per minute, which was close to normal speech rate. After improvement of the language model used for word recognition, the error rate was 12% for the vocabulary of 125,000 words. This was the first successful demonstration of a large word stock interpretation using neurotechnology. The source of a reliable signal, contrary to classical approaches, was not the Broca’s area (area 44), but the ventral premotor cortex (area v6). Based on the results of this study, the researchers identified two aspects of the neural code of speech, which were promising for the speech BCIs and were preserved years after the onset of paralysis: spatially-varied adjustment to speech articulators, which allowed accurate signal decoding from a small area of the cerebral cortex, and detailed articulatory representation of phonemes.
Conclusion
In recent years, one can see a continuous development of invasive neural interface technologies. However, while the development and research related to non-invasive BCIs is conducted in many countries, clinical studies of invasive BCIs, due to the need for significant funding, can only be conducted by a few research groups in the world. Each year their data enrich our understanding of the brain functioning and provide new capabilities for rehabilitation of patients with severe functional impairment. Such interdisciplinary achievements and research of invasive BCIs contribute to significant progress in development of both neurobiology and information technology.
Further advances here will be related to the increase of speed, accuracy, and multi-task BCI control by improving the design of neural implants and their biocompatibility; development of methods for obtaining biosignals and improvement of algorithms for their extraction and decoding [16, 91–100]; adjustment of the BCI design for home use [88, 89]. Also, BCI technologies are developed to restore locomotion [48] and use in pediatrics [101]; visual and auditory bionic prostheses are being designed [102]. Due to a gradual integration of BCI technologies into clinical practice, issues of standardization and bioethics shall unavoidably arise [103].
Study funding. The review was performed within the framework of the State Assignment of the Ministry of Health of the Russian Federation No.122051700017-2.
Conflicts of interest. There are no conflicts of interest.
References
- Mokienko O.A., Lyukmanov R.Kh., Bobrov P.D., Suponeva N.A., Piradov M.A. Brain–computer interfaces for upper limb motor recovery after stroke: current status and development prospects (review). Sovremennye tehnologii v medicine 2023; 15(6): 63, https://doi.org/10.17691/stm2023.15.6.07.
- Zhao Z.P., Nie C., Jiang C.T., Cao S.H., Tian K.X., Yu S., Gu J.W. Modulating brain activity with invasive brain-computer interface: a narrative review. Brain Sci 2023; 13(1): 134, https://doi.org/10.3390/brainsci13010134.
- Carvalho R., Dias N., Cerqueira J.J. Brain-machine interface of upper limb recovery in stroke patients rehabilitation: a systematic review. Physiother Res Int 2019; 24(2): e1764, https://doi.org/10.1002/pri.1764.
- Baniqued P.D.E., Stanyer E.C., Awais M., Alazmani A., Jackson A.E., Mon-Williams M.A., Mushtaq F., Holt R.J. Brain-computer interface robotics for hand rehabilitation after stroke: a systematic review. J Neuroeng Rehabil 2021; 18(1): 15, https://doi.org/10.1186/s12984-021-00820-8.
- Fu J., Chen S., Jia J. Sensorimotor rhythm-based brain-computer interfaces for motor tasks used in hand upper extremity rehabilitation after stroke: a systematic review. Brain Sci 2022; 13(1): 56, https://doi.org/10.3390/brainsci13010056.
- Bai Z., Fong K.N.K., Zhang J.J., Chan J., Ting K.H. Immediate and long-term effects of BCI-based rehabilitation of the upper extremity after stroke: a systematic review and meta-analysis. J Neuroeng Rehabil 2020; 17(1): 57, https://doi.org/10.1186/s12984-020-00686-2.
- Kruse A., Suica Z., Taeymans J., Schuster-Amft C. Effect of brain-computer interface training based on non-invasive electroencephalography using motor imagery on functional recovery after stroke — a systematic review and meta-analysis. BMC Neurol 2020; 20(1): 385, https://doi.org/10.1186/s12883-020-01960-5.
- Yang W., Zhang X., Li Z., Zhang Q., Xue C., Huai Y. The effect of brain-computer interface training on rehabilitation of upper limb dysfunction after stroke: a meta-analysis of randomized controlled trials. Front Neurosci 2021; 15: 766879, https://doi.org/10.3389/fnins.2021.766879.
- Mansour S., Ang K.K., Nair K.P.S., Phua K.S., Arvaneh M. Efficacy of brain-computer interface and the impact of its design characteristics on poststroke upper-limb rehabilitation: a systematic review and meta-analysis of randomized controlled trials. Clin EEG Neurosci 2022; 53(1): 79–90, https://doi.org/10.1177/15500594211009065.
- Peng Y., Wang J., Liu Z., Zhong L., Wen X., Wang P., Gong X., Liu H. The application of brain-computer interface in upper limb dysfunction after stroke: a systematic review and meta-analysis of randomized controlled trials. Front Hum Neurosci 2022; 16: 798883, https://doi.org/10.3389/fnhum.2022.798883.
- Nojima I., Sugata H., Takeuchi H., Mima T. Brain-computer interface training based on brain activity can induce motor recovery in patients with stroke: a meta-analysis. Neurorehabil Neural Repair 2022; 36(2): 83–96, https://doi.org/10.1177/15459683211062895.
- Xie Y.L., Yang Y.X., Jiang H., Duan X.Y., Gu L.J., Qing W., Zhang B., Wang Y.X. Brain-machine interface-based training for improving upper extremity function after stroke: a meta-analysis of randomized controlled trials. Front Neurosci 2022; 16: 949575, https://doi.org/10.3389/fnins.2022.949575.
- Shou Y.Z., Wang X.H., Yang G.F. Verum versus Sham brain-computer interface on upper limb function recovery after stroke: a systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore) 2023; 102(26): e34148, https://doi.org/10.1097/md.0000000000034148.
- Frolov A.A., Mokienko O., Lyukmanov R., Biryukova E., Kotov S., Turbina L., Nadareyshvily G., Bushkova Y. Post-stroke rehabilitation training with a motor-imagery-based brain-computer interface (BCI)-controlled hand exoskeleton: a randomized controlled multicenter trial. Front Neurosci 2017; 11: 400, https://doi.org/10.3389/fnins.2017.00400.
- Lyukmanov R.K., Isaev M.R., Mokienko O.A., Bobrov P.D., Ikonnikova E.S., Cherkasova A.N., Suponeva N.A. Brain–computer interface using functional near-infrared spectroscopy for post-stroke motor rehabilitation: case series. Ann Clin Exp Neurol 2023; 17(4): 82–88, https://doi.org/10.54101/acen.2023.4.10.
- Dong Y., Wang S., Huang Q., Berg RW., Li G., He J. Neural decoding for intracortical brain–computer interfaces. Cyborg Bionic Syst 2023; 4: 0044, https://doi.org/10.34133/cbsystems.0044.
- Vansteensel M.J., Pels E.G.M., Bleichner M.G., Branco M.P., Denison T., Freudenburg Z.V., Gosselaar P., Leinders S., Ottens T.H., Van Den Boom M.A., Van Rijen P.C., Aarnoutse E.J., Ramsey N.F. Fully implanted brain–computer interface in a locked-in patient with ALS. N Engl J Med 2016; 375(21): 2060–2066, https://doi.org/10.1056/nejmoa1608085.
- Hochberg L.R., Serruya M.D., Friehs G.M., Mukand J.A., Saleh M., Caplan A.H., Branner A., Chen D., Penn R.D., Donoghue J.P. Neuronal ensemble control of prosthetic devices by a human with tetraplegia. Nature 2006; 442(7099): 164–171, https://doi.org/10.1038/nature04970.
- Hochberg L.R., Bacher D., Jarosiewicz B., Masse N.Y., Simeral J.D., Vogel J., Haddadin S., Liu J., Cash S.S., van der Smagt P., Donoghue J.P. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 2012; 485(7398): 372–375, https://doi.org/10.1038/nature11076.
- Collinger J.L., Wodlinger B., Downey J.E., Wang W., Tyler-Kabara E.C., Weber D.J., McMorland A.J.C., Velliste M., Boninger M.L., Schwartz A.B. High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 2013; 381(9866): 557–564, https://doi.org/10.1016/s0140-6736(12)61816-9.
- Anumanchipalli G.K., Chartier J., Chang E.F. Speech synthesis from neural decoding of spoken sentences. Nature 2019; 568(7753): 493–498, https://doi.org/10.1038/s41586-019-1119-1.
- Willett F.R., Avansino D.T., Hochberg L.R., Henderson J.M., Shenoy K.V. High-performance brain-to-text communication via handwriting. Nature 2021; 593(7858): 249–254, https://doi.org/10.1038/s41586-021-03506-2.
- Flesher S.N., Downey J.E., Weiss J.M., Hughes C.L., Herrera A.J., Tyler-Kabara E.C., Boninger M.L., Collinger J.L., Gaunt R.A. A brain–computer interface that evokes tactile sensations improves robotic arm control. Science 2021; 372(6544): 831–836, https://doi.org/10.1126/science.abd0380.
- Willett F.R., Kunz E.M., Fan C., Avansino D.T., Wilson G.H., Choi E.Y., Kamdar F., Glasser M.F., Hochberg L.R., Druckmann S., Shenoy K.V., Henderson J.M. A high-performance speech neuroprosthesis. Nature 2023; 620(7976): 1031–1036, https://doi.org/10.1038/s41586-023-06377-x.
- Aflalo T., Kellis S., Klaes C., Lee B., Shi Y., Pejsa K., Shanfield K., Hayes-Jackson S., Aisen M., Heck C., Liu C., Andersen R.A. Neurophysiology. Decoding motor imagery from the posterior parietal cortex of a tetraplegic human. Science 2015; 348(6237): 906–910, https://doi.org/10.1126/science.aaa5417.
- Metzger S.L., Littlejohn K.T., Silva A.B., Moses D.A., Seaton M.P., Wang R., Dougherty M.E., Liu J.R., Wu P., Berger M.A., Zhuravleva I., Tu-Chan A., Ganguly K., Anumanchipalli G.K., Chang E.F. A high-performance neuroprosthesis for speech decoding and avatar control. Nature 2023; 620(7976): 1037–1046, https://doi.org/10.1038/s41586-023-06443-4.
- Ajiboye A.B., Willett F.R., Young D.R., Memberg W.D., Murphy B.A., Miller J.P., Walter B.L., Sweet J.A., Hoyen H.A., Keith M.W., Peckham P.H., Simeral J.D., Donoghue J.P., Hochberg L.R., Kirsch R.F. Restoration of reaching and grasping movements through brain-controlled muscle stimulation in a person with tetraplegia: a proof-of-concept demonstration. Lancet 2017; 389(10081): 1821–1830, https://doi.org/10.1016/s0140-6736(17)30601-3.
- Wang W., Collinger J.L., Degenhart A.D., Tyler-Kabara E.C., Schwartz A.B., Moran D.W., Weber D.J., Wodlinger B., Vinjamuri R.K., Ashmore R.C., Kelly J.W., Boninger M.L. An electrocorticographic brain interface in an individual with tetraplegia. PloS One 2013; 8(2): e55344, https://doi.org/10.1371/journal.pone.0055344.
- Davis K.C., Meschede-Krasa B., Cajigas I., Prins N.W., Alver C., Gallo S., Bhatia S., Abel J.H., Naeem J.A., Fisher L., Raza F., Rifai W.R., Morrison M., Ivan M.E., Brown E.N., Jagid J.R., Prasad A. Design-development of an at-home modular brain–computer interface (BCI) platform in a case study of cervical spinal cord injury. J Neuroeng Rehabil 2022; 19(1): 53, https://doi.org/10.1186/s12984-022-01026-2.
- Oxley T.J., Yoo P.E., Rind G.S., Ronayne S.M., Lee C.M.S., Bird C., Hampshire V., Sharma R.P., Morokoff A., Williams D.L., MacIsaac C., Howard M.E., Irving L., Vrljic I., Williams C., John S.E., Weissenborn F., Dazenko M., Balabanski A.H., Friedenberg D., Burkitt A.N., Wong Y.T., Drummond K.J., Desmond P., Weber D., Denison T., Hochberg L.R., Mathers S., O’Brien T.J., May C.N., Mocco J., Grayden D.B., Campbell B.C.V., Mitchell P., Opie N.L. Motor neuroprosthesis implanted with neurointerventional surgery improves capacity for activities of daily living tasks in severe paralysis: first in-human experience. J Neurointerv Surg 2021; 13(2): 102–108, https://doi.org/10.1136/neurintsurg-2020-016862.
- Mitchell P., Lee S.C.M., Yoo P.E., Morokoff A., Sharma R.P., Williams D.L., MacIsaac C., Howard M.E., Irving L., Vrljic I., Williams C., Bush S., Balabanski A.H., Drummond K.J., Desmond P., Weber D., Denison T., Mathers S., O’Brien T.J., Mocco J., Grayden D.B., Liebeskind D.S., Opie N.L., Oxley T.J., Campbell B.C.V. Assessment of safety of a fully implanted endovascular brain-computer interface for severe paralysis in 4 patients: the stentrode with thought-controlled digital switch (SWITCH) study. JAMA Neurol 2023; 80(3): 270–278, https://doi.org/10.1001/jamaneurol.2022.4847.
- Saha S., Mamun K.A., Ahmed K., Mostafa R., Naik G.R., Darvishi S., Khandoker A.H., Baumert M. Progress in brain computer interface: challenges and opportunities. Front Syst Neurosci 2021; 15: 578875, https://doi.org/10.3389/fnsys.2021.578875.
- Stavisky S.D., Willett F.R., Wilson G.H., Murphy B.A., Rezaii P., Avansino D.T., Memberg W.D., Miller J.P., Kirsch R.F., Hochberg L.R., Ajiboye A.B., Druckmann S., Shenoy K.V., Henderson J.M. Neural ensemble dynamics in dorsal motor cortex during speech in people with paralysis. ELife 2019; 8: e46015, https://doi.org/10.7554/elife.46015.
- Stavisky S.D., Willett F.R., Avansino D.T., Hochberg L.R., Shenoy K.V., Henderson J.M. Speech-related dorsal motor cortex activity does not interfere with iBCI cursor control. J Neural Eng 2020; 17(1): 016049, https://doi.org/10.1088/1741-2552/ab5b72.
- Humphrey D.R., Schmidt E.M., Thompson W.D. Predicting measures of motor performance from multiple cortical spike trains. Science 1970; 170(3959): 758–762, https://doi.org/10.1126/science.170.3959.758.
- Lilly J.C. Instantaneous relations between the activities of closely spaced zones on the cerebral cortex; electrical figures during responses and spontaneous activity. Am J Physiol 1954; 176(3): 493–504, https://doi.org/10.1152/ajplegacy.1954.176.3.493.
- Fetz E.E. Operant conditioning of cortical unit activity. Science 1969; 163(3870): 955–958, https://doi.org/10.1126/science.163.3870.955.
- Schmidt E.M. Single neuron recording from motor cortex as a possible source of signals for control of external devices. Ann Biomed Eng 1980; 8(4–6): 339–349, https://doi.org/10.1007/bf02363437.
- Ifft P.J., Shokur S., Li Z., Lebedev M.A., Nicolelis M.A.L. A brain-machine interface enables bimanual arm movements in monkeys. Sci Transl Med 2013; 5(210): 210ra154, https://doi.org/10.1126/scitranslmed.3006159.
- Chapin J.K., Moxon K.A., Markowitz R.S., Nicolelis M.A.L. Real-time control of a robot arm using simultaneously recorded neurons in the motor cortex. Nat Neurosci 1999; 2(7): 664–670, https://doi.org/10.1038/10223.
- Taylor D.M., Tillery S.I.H., Schwartz A.B. Direct cortical control of 3D neuroprosthetic devices. Science 2002; 296(5574): 1829–1832, https://doi.org/10.1126/science.1070291.
- Wessberg J., Stambaugh C.R., Kralik J.D., Beck P.D., Laubach M., Chapin J.K., Kim J., Biggs S.J., Srinivasan M.A., Nicolelis M.A.L. Real-time prediction of hand trajectory by ensembles of cortical neurons in primates. Nature 2000; 408(6810): 361–365, https://doi.org/10.1038/35042582.
- Carmena J.M., Lebedev M.A., Crist R.E., O’Doherty J.E., Santucci D.M., Dimitrov D.F., Patil P.G., Henriquez C.S., Nicolelis M.A.L. Learning to control a brain–machine interface for reaching and grasping by primates. PLoS Biol 2003; 1(2): E42, https://doi.org/10.1371/journal.pbio.0000042.
- Lebedev M.A., Carmena J.M., O’Doherty J.E., Zacksenhouse M., Henriquez C.S., Principe J.C., Nicolelis M.A.L. Cortical ensemble adaptation to represent velocity of an artificial actuator controlled by a brain-machine interface. J Neurosci 2005; 25(19): 4681–4693, https://doi.org/10.1523/jneurosci.4088-04.2005.
- O’Doherty J.E., Lebedev M.A., Hanson T.L., Fitzsimmons N.A., Nicolelis M.A.L. A brain-machine interface instructed by direct intracortical microstimulation. Front Integr Neurosci 2009; 3: 20, https://doi.org/10.3389/neuro.07.020.2009.
- Fitzsimmons N.A., Lebedev M.A., Peikon I.D., Nicolelis M.A.L. Extracting kinematic parameters for monkey bipedal walking from cortical neuronal ensemble activity. Front Integr Neurosci 2009; 3: 3, https://doi.org/10.3389/neuro.07.003.2009.
- Velliste M., Perel S., Spalding M.C., Whitford A.S., Schwartz A.B. Cortical control of a prosthetic arm for self-feeding. Nature 2008; 453(7198): 1098–1101, https://doi.org/10.1038/nature06996.
- Capogrosso M., Milekovic T., Borton D., Wagner F., Moraud E.M., Mignardot J.B., Buse N., Gandar J., Barraud Q., Xing D., Rey E., Duis S., Jianzhong Y., Ko W.K., Li Q., Detemple P., Denison T., Micera S., Bezard E., Bloch J., Courtine G. A brain-spine interface alleviating gait deficits after spinal cord injury in primates. Nature 2016; 539(7628): 284–288, https://doi.org/10.1038/nature20118.
- Ganguly K., Carmena J.M. Emergence of a stable cortical map for neuroprosthetic control. PLoS Biol 2009; 7(7): e1000153, https://doi.org/10.1371/journal.pbio.1000153.
- Gilja V., Nuyujukian P., Chestek C.A., Cunningham J.P., Yu B.M., Fan J.M., Churchland M.M., Kaufman M.T., Kao J.C., Ryu S.I., Shenoy K.V. A high-performance neural prosthesis enabled by control algorithm design. Nat Neurosci 2012; 15(12): 1752–1757, https://doi.org/10.1038/nn.3265.
- Ma X., Rizzoglio F., Bodkin K.L., Perreault E., Miller L.E., Kennedy A. Using adversarial networks to extend brain computer interface decoding accuracy over time. ELife 2023; 12: e84296, https://doi.org/10.7554/elife.84296.
- Athalye V.R., Khanna P., Gowda S., Orsborn A.L., Costa R.M., Carmena J.M. Invariant neural dynamics drive commands to control different movements. Curr Biol 2023; 33(14): 2962–2976.e15, https://doi.org/10.1016/j.cub.2023.06.027.
- Borra D., Filippini M., Ursino M., Fattori P., Magosso E. Motor decoding from the posterior parietal cortex using deep neural networks. J Neural Eng 2023; 20(3): 036016, https://doi.org/10.1088/1741-2552/acd1b6.
- Öztürk S., Devecioğlu İ., Güçlü B. Bayesian prediction of psychophysical detection responses from spike activity in the rat sensorimotor cortex. J Comput Neurosci 2023; 51(2): 207–222, https://doi.org/10.1007/s10827-023-00844-0.
- Orsborn A.L., Moorman H.G., Overduin S.A., Shanechi M.M., Dimitrov D.F., Carmena J.M. Closed-loop decoder adaptation shapes neural plasticity for skillful neuroprosthetic control. Neuron 2014; 82(6): 1380–1393, https://doi.org/10.1016/j.neuron.2014.04.048.
- Kao J.C., Nuyujukian P., Ryu S.I., Shenoy K.V. A high-performance neural prosthesis incorporating discrete state selection with hidden Markov models. IEEE Trans Biomed Eng 2017; 64(4): 935–945, https://doi.org/10.1109/tbme.2016.2582691.
- Willsey M.S., Nason-Tomaszewski S.R., Ensel S.R., Temmar H., Mender M.J., Costello J.T., Patil P.G., Chestek C.A. Real-time brain–machine interface in non-human primates achieves high-velocity prosthetic finger movements using a shallow feedforward neural network decoder. Nat Commun 2022; 13(1): 6899, https://doi.org/10.1038/s41467-022-34452-w.
- Levitskaya O.S., Lebedev M.A. Brain–computer interface: the future in the present. Vestnik Rossijskogo gosudarstvennogo medicinskogo universiteta 2016; 2: 4–16, https://doi.org/10.24075/brsmu.2016-02-01.
- Dennett D.C. A treat: Grey Walter’s precognitive carousel. In: Consciousness explained. New York, Boston, London: Back Bay Books/Little, Brown and Company, Hachette Book Group USA; 1991; p. 167–168.
- Kennedy P.R., Bakay R.A. Restoration of neural output from a paralyzed patient by a direct brain connection. Neuroreport 1998; 9(8): 1707–1711, https://doi.org/10.1097/00001756-199806010-00007.
- Kennedy P.R., Bakay R.A.E., Moore M.M., Adams K., Goldwaithe J. Direct control of a computer from the human central nervous system. IEEE Trans Rehabil Eng 2000; 8(2): 198–202, https://doi.org/10.1109/86.847815.
- Kennedy P.R., Kirby M.T., Moore M.M., King B., Mallory A. Computer control using human intracortical local field potentials. IEEE Trans Neural Syst Rehabil Eng 2004; 12(3): 339–344, https://doi.org/10.1109/tnsre.2004.834629.
- Bartels J., Andreasen D., Ehirim P., Mao H., Seibert S., Wright E.J., Kennedy P. Neurotrophic electrode: method of assembly and implantation into human motor speech cortex. J Neurosci Methods 2008; 174(2): 168–176, https://doi.org/10.1016/j.jneumeth.2008.06.030.
- Guenther F.H., Brumberg J.S., Wright E.J., Nieto-Castanon A., Tourville J.A., Panko M., Law R., Siebert S.A., Bartels J.L., Andreasen D.S., Ehirim P., Mao H., Kennedy P.R. A wireless brain-machine interface for real-time speech synthesis. PloS One 2009; 4(12): e8218, https://doi.org/10.1371/journal.pone.0008218.
- Brumberg J.S., Nieto-Castanon A., Kennedy P.R., Guenther F.H. Brain–computer interfaces for speech communication. Speech Commun 2010; 52(4): 367–379, https://doi.org/10.1016/j.specom.2010.01.001.
- Lebedev M.A. Brain-computer interface for the augmentation of brain functions. Nauka i innovacii v medicine 2016; 1(3): 11–27, https://doi.org/10.35693/2500-1388-2016-0-3-11-27.
- Maynard E.M., Nordhausen C.T., Normann R.A. The Utah intracortical electrode array: a recording structure for potential brain-computer interfaces. Electroencephalogr Clin Neurophysiol 1997; 102(3): 228–239, https://doi.org/10.1016/s0013-4694(96)95176-0.
- Guillory K.S., Normann R.A. A 100-channel system for real time detection and storage of extracellular spike waveforms. J Neurosci Methods 1999; 91(1–2): 21–29, https://doi.org/10.1016/s0165-0270(99)00076-x.
- Suner S., Fellows M.R., Vargas-Irwin C., Nakata G.K., Donoghue J.P. Reliability of signals from a chronically implanted, silicon-based electrode array in non-human primate primary motor cortex. IEEE Trans Neural Syst Rehabil Eng 2005; 13(4): 524–541, https://doi.org/10.1109/tnsre.2005.857687.
- Serruya M.D., Hatsopoulos N.G., Paninski L., Fellows M.R., Donoghue J.P. Instant neural control of a movement signal. Nature 2002; 416(6877): 141–142, https://doi.org/10.1038/416141a.
- Donoghue J.P., Nurmikko A., Black M., Hochberg L.R. Assistive technology and robotic control using motor cortex ensemble-based neural interface systems in humans with tetraplegia. J Physiol 2007; 579(Pt 3): 603–611, https://doi.org/10.1113/jphysiol.2006.127209.
- Kim S.P., Simeral J.D., Hochberg L.R., Donoghue J.P., Friehs G.M., Black M.J. Point-and-click cursor control with an intracortical neural interface system by humans with tetraplegia. IEEE Trans Neural Syst Rehabil Eng 2011; 19(2): 193–203, https://doi.org/10.1109/tnsre.2011.2107750.
- Truccolo W., Friehs G.M., Donoghue J.P., Hochberg L.R. Primary motor cortex tuning to intended movement kinematics in humans with tetraplegia. J Neurosci 2008; 28(5): 1163–1178, https://doi.org/10.1523/jneurosci.4415-07.2008.
- Kim S.P., Simeral J.D., Hochberg L.R., Donoghue J.P., Black M.J. Neural control of computer cursor velocity by decoding motor cortical spiking activity in humans with tetraplegia. J Neural Eng 2008; 5(4): 455–476, https://doi.org/10.1088/1741-2560/5/4/010.
- Simeral J.D., Kim S.P., Black M.J., Donoghue J.P., Hochberg L.R. Neural control of cursor trajectory and click by a human with tetraplegia 1000 days after implant of an intracortical microelectrode array. J Neural Eng 2011; 8(2): 025027, https://doi.org/10.1088/1741-2560/8/2/025027.
- Wodlinger B., Downey J.E., Tyler-Kabara E.C., Schwartz A.B., Boninger M.L., Collinger J.L. Ten-dimensional anthropomorphic arm control in a human brain-machine interface: difficulties, solutions, and limitations. J Neural Eng 2015; 12(1): 016011, https://doi.org/10.1088/1741-2560/12/1/016011.
- Downey J.E., Brane L., Gaunt R.A., Tyler-Kabara E.C., Boninger M.L., Collinger J.L. Motor cortical activity changes during neuroprosthetic-controlled object interaction. Sci Rep 2017; 7(1): 16947, https://doi.org/10.1038/s41598-017-17222-3.
- Handelman D.A., Osborn L.E., Thomas T.M., Badger A.R., Thompson M., Nickl R.W., Anaya M.A., Wormley J.M., Cantarero G.L., McMullen D., Crone N.E., Wester B., Celnik P.A., Fifer M.S., Tenore F.V. Shared control of bimanual robotic limbs with a brain-machine interface for self-feeding. Front Neurorobot 2022; 16: 918001, https://doi.org/10.3389/fnbot.2022.918001.
- Gilja V., Pandarinath C., Blabe C.H., Nuyujukian P., Simeral J.D., Sarma A.A., Sorice B.L., Perge J.A., Jarosiewicz B., Hochberg L.R., Shenoy K.V., Henderson J.M. Clinical translation of a high-performance neural prosthesis. Nat Med 2015; 21(10): 1142–1145, https://doi.org/10.1038/nm.3953.
- Brandman D.M., Hosman T., Saab J., Burkhart M.C., Shanahan B.E., Ciancibello J.G., Sarma A.A., Milstein D.J., Vargas-Irwin C.E., Franco B., Kelemen J., Blabe C., Murphy B.A., Young D.R., Willett F.R., Pandarinath C., Stavisky S.D., Kirsch R.F., Walter B.L., Bolu Ajiboye A., Cash S.S., Eskandar E.N., Miller J.P., Sweet J.A., Shenoy K.V., Henderson J.M., Jarosiewicz B., Harrison M.T., Simeral J.D., Hochberg L.R. Rapid calibration of an intracortical brain-computer interface for people with tetraplegia. J Neural Eng 2018; 15(2): 026007, https://doi.org/10.1088/1741-2552/aa9ee7.
- Paulo D.L., Sheth S.A., McKhann G.M. II. Living brain to hand: bypassing spinal cord quadriplegia using a novel neuroprosthesis. Neurosurgery 2016; 79(2): N13–N14, https://doi.org/10.1227/01.neu.0000489885.18088.eb.
- Chadwick E.K., Blana D., Simeral J.D., Lambrecht J., Kim S.P., Cornwell A.S., Taylor D.M., Hochberg L.R., Donoghue J.P., Kirsch R.F. Continuous neuronal ensemble control of simulated arm reaching by a human with tetraplegia. J Neural Eng 2011; 8(3): 034003, https://doi.org/10.1088/1741-2560/8/3/034003.
- Bouton C.E., Shaikhouni A., Annetta N.V., Bockbrader M.A., Friedenberg D.A., Nielson D.M., Sharma G., Sederberg P.B., Glenn B.C., Mysiw W.J., Morgan A.G., Deogaonkar M., Rezai A.R. Restoring cortical control of functional movement in a human with quadriplegia. Nature 2016; 533(7602): 247–250, https://doi.org/10.1038/nature17435.
- Hughes C., Herrera A., Gaunt R., Collinger J. Bidirectional brain-computer interfaces. Handb Clin Neurol 2020; 168: 163–181, https://doi.org/10.1016/B978-0-444-63934-9.00013-5.
- Bacher D., Jarosiewicz B., Masse N.Y., Stavisky S.D., Simeral J.D., Newell K., Oakley E.M., Cash S.S., Friehs G., Hochberg L.R. Neural point-and-click communication by a person with incomplete locked-in syndrome. Neurorehabil Neural Repair 2015; 29(5): 462–471, https://doi.org/10.1177/1545968314554624.
- Jarosiewicz B., Sarma A.A., Bacher D., Masse N.Y., Simeral J.D., Sorice B., Oakley E.M., Blabe C., Pandarinath C., Gilja V., Cash S.S., Eskandar E.N., Friehs G., Henderson J.M., Shenoy K.V., Donoghue J.P., Hochberg L.R. Virtual typing by people with tetraplegia using a self-calibrating intracortical brain-computer interface. Sci Transl Med 2015; 7(313): 313ra179, https://doi.org/10.1126/scitranslmed.aac7328.
- Pandarinath C., Nuyujukian P., Blabe C.H., Sorice B.L., Saab J., Willett F.R., Hochberg L.R., Shenoy K.V., Henderson J.M. High performance communication by people with paralysis using an intracortical brain-computer interface. ELife 2017; 6: e18554, https://doi.org/10.7554/elife.18554.
- Nuyujukian P., Albites Sanabria J., Saab J., Pandarinath C., Jarosiewicz B., Blabe C.H., Franco B., Mernoff S.T., Eskandar E.N., Simeral J.D., Hochberg L.R., Shenoy K.V., Henderson J.M. Cortical control of a tablet computer by people with paralysis. PloS One 2018; 13(11): e0204566, https://doi.org/10.1371/journal.pone.0204566.
- Simeral J.D., Hosman T., Saab J., Flesher S.N., Vilela M., Franco B., Kelemen J.N., Brandman D.M., Ciancibello J.G., Rezaii P.G., Eskandar E.N., Rosler D.M., Shenoy K.V., Henderson J.M., Nurmikko A.V., Hochberg L.R. Home use of a percutaneous wireless intracortical brain-computer interface by individuals with tetraplegia. IEEE Trans Biomed Eng 2021; 68(7): 2313–2325, https://doi.org/10.1109/tbme.2021.3069119.
- Shah N.P., Willsey M.S., Hahn N., Kamdar F., Avansino D.T., Hochberg L.R., Shenoy K.V., Henderson J.M. A brain-computer typing interface using finger movements. Int IEEE EMBS Conf Neural Eng 2023; 2023: 10.1109/ner52421.2023.10123912, https://doi.org/10.1109/ner52421.2023.10123912.
- Dekleva B.M., Weiss J.M., Boninger M.L., Collinger J.L. Generalizable cursor click decoding using grasp-related neural transients. J Neural Eng 2021; 18(4): 0460e9, https://doi.org/10.1088/1741-2552/ac16b2.
- Jun J.J., Steinmetz N.A., Siegle J.H., Denman D.J., Bauza M., Barbarits B., Lee A.K., Anastassiou C.A., Andrei A., Aydın Ç., Barbic M., Blanche T.J., Bonin V., Couto J., Dutta B., Gratiy S.L., Gutnisky D.A., Häusser M., Karsh B., Ledochowitsch P., Lopez C.M., Mitelut C., Musa S., Okun M., Pachitariu M., Putzeys J., Rich P.D., Rossant C., Sun W.L., Svoboda K., Carandini M., Harris K.D., Koch C., O’Keefe J., Harris T.D. Fully integrated silicon probes for high-density recording of neural activity. Nature 2017; 551(7679): 232–236, https://doi.org/10.1038/nature24636.
- Musk E.; Neuralink. An integrated brain-machine interface platform with thousands of channels. J Med Internet Res 2019; 21(10): e16194, https://doi.org/10.2196/16194.
- Nurmikko A. Challenges for large-scale cortical interfaces. Neuron 2020; 108(2): 259–269, https://doi.org/10.1016/j.neuron.2020.10.015.
- Vázquez-Guardado A., Yang Y., Bandodkar A.J., Rogers J.A. Recent advances in neurotechnologies with broad potential for neuroscience research. Nat Neurosci 2020; 23(12): 1522–1536, https://doi.org/10.1038/s41593-020-00739-8.
- Sahasrabuddhe K., Khan A.A., Singh A.P., Stern T.M., Ng Y., Tadić A., Orel P., LaReau C., Pouzzner D., Nishimura K., Boergens K.M., Shivakumar S., Hopper M.S., Kerr B., Hanna M.E.S., Edgington R.J., McNamara I., Fell D., Gao P., Babaie-Fishani A., Veijalainen S., Klekachev A.V., Stuckey A.M., Luyssaert B., Kozai T.D.Y., Xie C., Gilja V., Dierickx B., Kong Y., Straka M., Sohal H.S., Angle M.R. The Argo: a 65,536 channel recording system for high density neural recording in vivo. BioRxiv; 2020, https://doi.org/10.1101/2020.07.17.209403.
- Zhou Y., Yang H., Wang X., Yang H., Sun K., Zhou Z., Sun L., Zhao J., Tao T.H., Wei X. A mosquito mouthpart-like bionic neural probe. Microsyst Nanoeng 2023; 9: 88, https://doi.org/10.1038/s41378-023-00565-5.
- Trotier A., Bagnoli E., Walski T., Evers J., Pugliese E., Lowery M., Kilcoyne M., Fitzgerald U., Biggs M. Micromotion derived fluid shear stress mediates peri-electrode gliosis through mechanosensitive ion channels. Adv Sci (Weinh) 2023; 10(27): e2301352, https://doi.org/10.1002/advs.202301352.
- Petrosyan A., Voskoboinikov A., Sukhinin D., Makarova A., Skalnaya A., Arkhipova N., Sinkin M., Ossadtchi A. Speech decoding from a small set of spatially segregated minimally invasive intracranial EEG electrodes with a compact and interpretable neural network. J Neural Eng 2022; 19(6): 066016, https://doi.org/10.1088/1741-2552/aca1e1.
- Chen K., Cambi F., Kozai T.D.Y. Pro-myelinating clemastine administration improves recording performance of chronically implanted microelectrodes and nearby neuronal health. Biomaterials 2023; 301: 122210, https://doi.org/10.1016/j.biomaterials.2023.122210.
- Bergeron D., Iorio-Morin C., Bonizzato M., Lajoie G., Orr Gaucher N., Racine É., Weil A.G. Use of invasive brain-computer interfaces in pediatric neurosurgery: technical and ethical considerations. J Child Neurol 2023; 38(3–4): 223–238, https://doi.org/10.1177/08830738231167736.
- Nowik K., Langwińska-Wośko E., Skopiński P., Nowik K.E., Szaflik J.P. Bionic eye review — an update. J Clin Neurosci 2020; 78: 8–19, https://doi.org/10.1016/j.jocn.2020.05.041.
- Drew L. The rise of brain-reading technology: what you need to know. Nature 2023; 623(7986): 241–243, https://doi.org/10.1038/d41586-023-03423-6.










