Estimation of General Toxicity and Immunological Safety of a Novel Therapeutic Vaccine Against Human Papillomavirus-Assosiated Diseases
The aim of the investigation was to study in experiment total toxicity and immunological safety of a novel domestic therapeutic vaccine against recurrent respiratory papillomatosis and anogenital condylomatosis, the vaccine injected intramuscularly.
Materials and Methods. We studied acute toxicity of the vaccine by the following parameters: clinical presentation of intoxication, median lethal dose size, the change of body weight of the surviving animals; chronic toxicity — by dynamics of general condition of the animals, body weight, hematological and biochemical indices of peripheral blood, functional status of central nervous system, cardiovascular system, kidneys, as well as pathomorphological changes of viscera.
Allergenicity was studied by systemic and active cutaneous anaphylactic tests. We assessed immunotoxicity by direct a hemagglutination assay and a delayed-type hypersensitivity test, as well as by neutrophilic activity using luminal-dependent chemiluminescence. Proliferative activity of В- and Т-lymphocytes to lipopolysaccharides and concanavalin А (ConА) was estimated in a direct immunofluorescence test using immunocytochemical assay with anti-Ki-67 monoclonal antibodies.
Results. Mean lethal doses (LD50) were not reached in white outbread rats injected intramuscularly by the tested vaccine at a maximum possible single dose (25 ml/kg). Multi-dose administration to white outbread rats and chinchilla rabbits at doses of 0.043; 0.43; 0.86 ml/kg and 0.023; 0.23; 0.4 ml/kg respectively, did not cause significant damage of functional status of basal organs and systems of experimental animals.
The findings of systemic anaphylaxis and active cutaneous anaphylactic response to the vaccine in albino guinea pigs at the doses of 0.033 and 0.33 ml/kg intramuscularly indicated the vaccine to exhibit no allergenic properties.
Intramuscular vaccine injected to first filial hybrid mice (СВА×С57BL/6)F1 at the doses of 0.084; 2.5 and 25 ml/kg showed neither significant increase or decrease of inflammatory response intensity relating to reference values. The vaccine at the doses of 0.084 and 2.5 ml/kg did not cause any changes of vaccination titer (IgG), phagocytic activity, spontaneous or induced blastic transformation of lymphocytes compared to the control group. The vaccine administered at the dose of 25 ml/kg significantly reduced the antibody formation level relating to the controls, as well as produced a suppressive effect on spontaneous and induced phagocytic activity resulting in the significant reduction of ConА-induced proliferative activity of Т-lymphocytes by the number of Ki-67 positive cells compared to the control group.
Conclusion. The animal study of different types of general toxicity of the vaccine against recurrent respiratory papillomatosis and anogenital condylomatosis, the vaccine injected intramuscularly, showed the vaccine to be low toxic and immunologically safe in the dose range from 0.033 to 2.5 ml/kg.
Human papilloma viruses (HPV), types 6 and 11, refer to viruses with low oncogenic risk and are associated with such diseases as anogenital condylomatosis and recurrent respiratory papillomatosis [1, 2]. The action of immunoprophylactic HPV vaccines — Gardasil® (Merck & Co, USA) and Cervarix® (GlaxoSmithKline Biologicals, Belgium) — aims at developing specific humoral immunity to capsid proteins of the virus (L-protein) resulting in infection prevention. However, a pharmaceutical market lacks specific medications to treat HPV-associated pathology [1, 2]. Therapeutic vaccines against HPV-associated diseases should target specific immunotherapy of chronic infections in patients by activating a cellular component of immunity, in particular, cytotoxic response of T-lymphocytes [3].
Research Institute for Genetics and Selection of Industrial Microorganisms has developed an innovative therapeutic vaccine against recurrent respiratory papillomatosis and anogenital condylomatosis based on hybrid recombinant proteins — oncoproteins Е7 HPV (types 6 and 11) cross-linked with heat shock proteins 70 Mycobacterium tuberculosis. Saccharomyces cerevisiae were used as a target recombinant protein producer [4].
The selection of an early oncoprotein Е7 as a target was due to high conservatism of amino acid sequence of the protein, its key role in life cycle of the virus, and the ability to express constitutively in epithelial cells affected by virus [5]. Heat shock protein is needed to enhance immunogenicity of a recombinant antigen by activating antigen-presenting cells and participation in antigen processing [6].
Within the framework of pharmaceutical development we are to assess general toxicity and immunologic safety of the vaccine as part of biological standartization and its quality control. The study of a nonspecific effect of the vaccine under development on immune system of animals is a compulsory part of preclinical studies of an innovator drug [7, 8]. In addition, we should take into consideration that the presence of residual of protein strain-producer of final dosage form of aluminium hydroxide sorbent as an activator of innate immune response system can result in allergic reactions [8–10].
The aim of the investigation was to study in experiment systemic toxicity and immunological safety of a novel domestic therapeutic vaccine against recurrent respiratory papillomatosis and anogenital condylomatosis, the vaccine injected intramuscularly.
Materials and Methods. The study was carried out in accordance with the European Convention for Protection of Vertebrate Animals Used with Experimental and Other Scientific Purposes (the Convention took place in Strasbourg on March, 18, 1986 and was confirmed in Strasbourg on June, 15, 2006) and approved by Ethics Committee of Nizhny Novgorod State Medical Academy.
A therapeutic vaccine against recurrent respiratory papillomatosis and anogenital condylomatosis is a sterile suspension for injections, one dose (0.5 ml) containing hybrid recombinant proteins — oncoprotein Е7 HPV, types 6 and 11, and heat shock protein 70 Mycobacterium tuberculosis (200 μg) sorbed on aluminium hydroxide (no more than aluminium, 1.25 mg per dose).
For experiments we used clinically healthy animals kept under similar housing and nutrition conditions. The animals were received from Research Center for Biomedical Technologies of Russian Academy of Medical Sciences, “Andreevka”.
Acute toxicity was assessed on white outbred mice and rats of both genders, the number of animals in each group being 12.
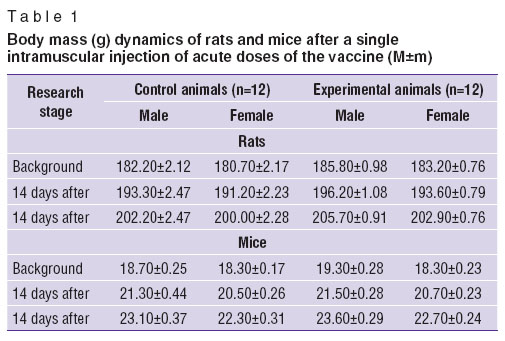
The doses tested ranged from 5 to 25 ml/kg calculated by the pharmaceutical form. Maximum volume for intramuscular injection was 0.5 ml/20 g body weight — for mice, and 5.0 ml/200 g — for rats. To achieve the necessary volumes and for the purposes of quality control we used sterile normal saline. We described clinical presentation of intoxication developed within 4 h after a single maximum dosage of vaccine. Moreover, we weighted the animals on day 7 and 14 after one dosage delivery of vaccine. After the experiment all animals were sacrificed by putting in СО2-chamber followed by decapitation to determine the mass coefficients of internal organs and their pathomorphological study.
Chronic toxicity was studied on white outbred rats and chinchilla rabbits of both genders, the number of animals being 20 and 12 respectively. We chose a mean therapeutic dose, which is to be clinically used. We converted human doses into animal ones using the method of equi-therapeutic doses considering body surface coefficient [7]. The doses of 0.043; 0.43 and 0.86 ml/kg were injected to rats intramuscularly; and the doses of 0.023; 0.23 and 0.4 ml/kg — to rabbits. The vaccine was administered in experimental groups, and normal saline — in control groups a day later, for 90 days.
To assess chronic toxicity we recorded general state of the animals, body mass dynamics. On days 30, 60, 90, and 30 days after the vaccine withdrawal we estimated hematological and biochemical parameters of peripheral blood, functional state of central nervous and cardiovascular systems, kidneys, as well as carried out a pathomorphological study of internal organs.
In accordance with domestic recommendations, immunologic safety study program involves allergenicity and immunotoxicity assessment including the estimation of humoral, cellular and nonspecific immune response. According to international requirements, in addition, the program includes the assessment of proliferative activity of В- and Т-lymphocytes to lipopolysaccharides (LPS) and concanavalin А (ConA) [10].
Allergenicity was studied on albino guinea pigs of both genders using systemic and active cutaneous anaphylactic tests. There were 10 animals in each group. Experimental animals were administered the test vaccine intramuscularly at an equi-therapeuticdose (0.033 ml/kg) and at the dose 10 times as much (0.33 ml/kg), according to the scheme described in the application guide [7]. As a negative test control we used sterile normal saline, as a positive test control — 0.6% albumen solution to assess systemic anaphylaxis, and 2,4-dinitrochlorbenzene (2,4-DNCB) — to assess active cutaneous anaphylaxis.
Immunotoxicity was studied on first filial hybrid mice of both genders (СВА×С57BL/6)F1 at doses 0.084; 2.5 and 25 ml/kg. The number of animals in the control and experimental group was 10. Humoral immune response was assessed by determining serum antibody titer (IgG) in direct hemagglutination reaction with Т-dependent antigen (sheep red blood cells) [7]. In the experiment, control mice and those of the experimental group were immunized intraperitoneally by sheep red blood cells (6·106/ml). Serum of immunized animals was obtained 7 days after intramuscular injection of the test vaccine at the doses indicated (normal saline was injected to control animals) followed by double dilution of 0.2 ml, starting with 1:5 dilution.
We assessed cell-mediated response by delayed-type hypersensitivity test to particulate antigen and hapten.In experimental groups, 1 h after a sensitizing dose (1·107/100 μl of sheep red blood cells) injected subcutaneously in interscapular region, or 0.2 ml 10 mM of 2,4,6-trinitrobenzenesulfonic acid (TNBS) in tail, as well as we administered the vaccine for the following 4 days intramuscularly at the doses indicated. Control animals were injected sterile normal saline, 0.5 ml, intramuscularly. A booster dose — 1·108/20 μl of sheep red blood cells — was injected intraplantarly on day 5 after sensibilization (or 0.02 ml 10 mM of TNBS solution on day after sensibilization). We registered the intensity of an inflammatory response 24 h after booster dose administration by measuring the mean diameter of edema area on a paw. We determined the inflammatory response intensity by the difference between the edema diameter in the animals with antigen injection and those administered sterile phosphate buffered saline (pH=7.4).
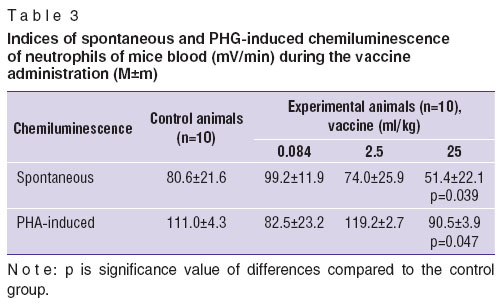
Nonspecific immune response was assessed by reactive changes of blood neutrophils, in particular, by the parameters of metabolic cell activation registered by luminal-dependent chemiluminescence. For phagocytic activity assessment we used heparinized blood of mice 24 h after a single intramuscular injection of the vaccine at the doses indicated. Heparinized blood was diluted in the ratio 1:100 by Hanks' solution (without phenol red). For chemiluminescence stimulation we used luminal solution (Chemapol, Czech Republic). The findings were recorded by a chemiluminometer Luminoskan Ascent (Thermo Scientific, Switzerland) at 37°С. Phagocytic activity was estimated by the parameters of spontaneous and phytohemagglutinin (PHG) induced chemiluminescence [11].
Functional activity of Т- and В-lymphocytes indicating the intensity of specific body sensitization was assessed by lymphocyte blastogenic response (LBR): T-lymphocyte — using ConA mitogen, B-lymphocyte — using LPS. Splenocyte proliferation in lymphocyte was assessed by means of imunicytochemistry using as a label a core antigen revealed in direct fluorescence using monoclonal antibodies (MAb) against protein Ki-67 [12]. The experimental animals were injected vaccine intramuscularly during 4 days at the doses indicated. Control animals were administered normal saline, 0.5 ml, intramuscularly. An hour after the last injection the animals were sacrificed in the above mentioned manner, their spleen was removed aseptically, the cell-rich fluid being prepared. Spleens were homogenized in medium 199, 5 ml (Microgene, Russia) with 1% fetal calf serum (FCS) solution. Obtained splenocytes were washed twice by medium 199 with 1% FCS solution. The reaction was carried out on the model of both induced and spontaneous blastogenic response with monoclonal antibodies to phycoerythrin-labeled (Sigma, USA) Ki-67 diluted in the ratio 1:20. The preparations obtained were studied using a fluorescent device Cell-Q (Carl Zeiss, Germany) counting the cells with luminescent foci in the nuclear area, and by means of a preliminary estimation of cell count using phase-contrast microscopy (DM1000, Leica, Germany).
The findings were statistically processed using a statistic package of Statistica 5.5. Statistical significance between the samplings with normal distribution was determined by Student t-test; between the samplings with non-normal distribution — using Wilcoxon test and Mann–Whitney U-test. The results obtained were presented as M±m, where M is arithmetic mean, m is standard error of the mean.
Results and Discussion. When studying acute toxicity, median lethal doses (LD50) in white outbred rats and mice (even if a maximum possible single dose of 25 ml/kg was administered) were not reached. It is evidence that a single dose of the test vaccine is low toxic. In addition, experimental animals were gaining weight during the observation period. No differences in weight gain were found compared to the control animals (Table 1). We recorded no visible differences in locomotor and eating behavior, in the state of integmentum and visible mucosa in their response to internal stimuli. According to morphological analysis of the organs under study, no differences between experimental and control animals were found either. The skin area (femoral muscle) the vaccine was injected had neither erythema, necrosis, edema, nor other changes of tissues.
|
|
The results of chronic toxicity study of the vaccine showed that multiple-dose administration of the test vaccine in the doses indicated caused no significant alterations in functional state of major organs and body systems. There was just significant transient increase of mass coefficients of thymus and ovaries in animals administered the vaccine for a long period of time at the dose of 0.86 ml/kg (for rats) and 0.4 ml/kg (for rabbits).
The findings of systemic anaphylaxis on the vaccine at the doses of 0.033 and 0.33 ml/kg indicated no allergenic properties (Table 2). It may be due to the fact that the vaccine mode of action is primarily related to Th1-lymphocyte stimulation that explains the absence of severe allergic reactions in vaccine administration.
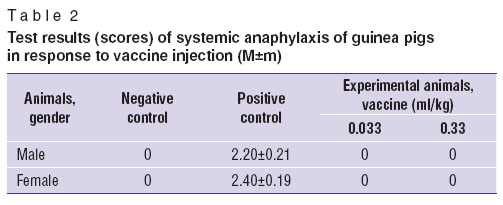 Table 2. Test results (scores) of systemic anaphylaxis of guinea pigs in response to vaccine injection (М±m) Table 2. Test results (scores) of systemic anaphylaxis of guinea pigs in response to vaccine injection (М±m)
|
Both male and female animals showed no active cutaneous anaphylactic reaction on the vacine at test doses (0.033 and 0.33 ml/kg). The spot diameter in experimental groups and the groups of negative control did not exceed 0.28±0.12 mm against high-grade sensitivity to 1% solution of 2,4-DNCB (8.32±0.26 mm).
The study of humoral immune response showed that the vaccine at the doses 0.084 and 2.5 ml/kg did not cause any significant changes of vaccination titer (IgG) (32.0±4.2 and 26.4±3.9 respectively) compared to the control group (33.6±5.9). The dose of 25 ml/kg (0.5 ml per mouse) resulted in significant decrease of antibody formation (titer 19.2±3.6) relating to the control group (33.6±5.9; p=0.045) that may be due to large doses of aluminium hydroxide comprising the vaccine.
The study of the test vaccine effect on cell-mediated response demonstrated that vaccine administration in all groups of experimental animals caused no significant increase or decrease of inflammatory response intensity in relation to the reference values. It is likely to be due to low production of proinflammatory cytokines in response to the vaccine injection.
When studying nonspecific immune response, the findings of spontaneous chemiluminescence showed significant activity decrease of phagocytes when the vaccine was injected only at the dose of 25 ml/kg (0.5 ml per mouse) relating to the control group (Table 3).The results of induced chemiluminescence showed that neutrophil reactivity, i.e. the ability of cells to respond to stimuli significantly decreased as well in relation to reference values only against the administration of 25-ml/kg dose (0.5 ml per mouse).
|
|
The findings suggest that the administration of the vaccine at the doses 0.084 and 2.5 ml/kg has no effect on phagocytosis activity, while the dose of 25 ml/kg (0.5 ml per mouse) has a suppressive effect on spontaneous and induced phagocytic activity.
The study of functional activity of Т- and В-lymphocytes showed that vaccine administered at the doses of 0.084 and 2.5 ml/kg did not result in significant changes of spontaneous and induced blastic transformation of splenocytes compared to the control group (Table 4). However, after the injection of 25 ml/kg (0.5 ml per mouse) there was significant decrease of ConA-induced proliferative activity by the number of Ki-67 positive cells compared to the control group indicating reduced intensity of specific sensibilization of the body if the vaccine is administered at the dose indicated.
|
|
The findings can be used to obtain permission for carrying out clinical trials of efficiency and safety of the therapeutical vaccine under study.
Conclusion. The study of general toxicity and immunologic safety of a novel domestic vaccine to treat HPV-associated diseases (recurrent respiratory papillomatosis and anogenital condylomatosis) suggests that the vaccine is low toxic, and immunologically safe — in the dose range from 0.033 to 2.5 ml/kg (for experimental animals of different types). Vaccine injection at the dose of 25 ml/kg has a suppressive effect on humoral immune response, phagocytic activity, as well as reduces mitogen-induced proliferation of Т-lymphocytes.
Study Funding. The study was financially supported by Ministry of Education and Science of the Russian Federation within the frame of government contract No.16.N08.12.1024 dated June, 14, 2012 “Preclinical studies of domestic therapeutic vaccine based on eukaryote cells against recurrent respiratory papillomatosis and anogenital condylomatosis”.
Conflicts of Interest. The authors have no conflicts of interest.
References
- Barr E., Sings H.L. Prophylactic HPV vaccines: new interventions for cancer control. Vaccine 2008 Nov 18; 26(49): 6244–6257, http://dx.doi.org/10.1016/j.vaccine.2008.07.056.
- Kahn J.A. Vaccination as a prevention strategy for human papillomavirus-related diseases. J Adolesc Health 2005 Dec; 37(6 Suppl): S10–S16, http://dx.doi.org/10.1016/j.jadohealth.2005.08.018.
- Mazitova L.P., Aslamazian L.K., Namazova L.S., Shaipov T.S. Peculiarities of clinical treatment, diagnostics and approaches to papilloma viral therapy in childhood. Pediatricheskaya farmakologiya 2006; 3(6): 51–54.
- Kozlov D.G., Cheperegin S.E., Gubaydullin I.I., Efremov B.D., Tyurin O.V., Zalunin I.A. Sposob polucheniya belka E7-HSP70 i shtamm drozhzhey Saccharomyces cerevisiae dlya ego osushchestvleniya [Method for Е7-HSP70 producion, and Saccharomyces cerevisiae yeast strain for its implementation]. Patent RF 2489481. 2013.
- Chu N.R., Wu H.B., Wu T., Boux L.J., Siegel M.I., Mizzen L.A. Immunotherapy of a human papillomavirus (HPV) type 16 E7-expressing tumour by administration of fusion protein comprising Mycobacterium bovis bacille Calmette-Guérin (BCG) HSP65 and HPV16 E7. Clin Exp Immunol 2000 Aug; 121(2): 216–225, http://dx.doi.org/10.1046/j.1365-2249.2000.01293.x.
- Chu N.R. Therapeutic vaccination for the treatment of mucosotropic human papillomavirus-associated disease. Expert Opin Biol Ther 2003 Jun; 3(3): 477–486, http://dx.doi.org/10.1517/14712598.3.3.477.
- Rukovodstvo po provedeniyu doklinicheskikh issledovaniy lekarstvennykh sredstv. Ch. 1 [Manual on conducting preclinical drug trials. Part 1]. Pod red. Mironova A.N. [Mironov A.N. (editor)]. Moscow: Grif i K; 2012; 944 p.
- ICH guideline S6 (R1) — preclinical safety evaluation of biotechnology-derived pharmaceuticals. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500002828.pdf.
- Urakov A.L. Osnovy klinicheskoy farmakologii [Basic clinical pharmacology]. Izhevsk: Izhevskiy poligrafkombinat; 1997; 164 p.
- Note for guidance on repeated dose toxicity. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003102.pdf.
- Allen R.C. Chemiluminescence and the study of phagocyte redox metabolism. Adv Exp Med Biol 1982; 141: 411–421, http://dx.doi.org/10.1007/978-1-4684-8088-7_39.
- Gerdes J., Schwab U., Lemke H., Stein H. Production of a mouse monoclonal antibody reactive with a human nuclear antigen associated with cell proliferation. Int J Cancer 1983 Jan 15; 31(1): 13–20, http://dx.doi.org/10.1002/ijc.2910310104.