Biodistribution of Amine-Amide Chlorin e6 Derivative Conjugate with a Boron Nanoparticle for Boron Neutron-Capture Therapy
The aim of the investigation was to study the biodistribution of amino-amide chlorin e6 derivative conjugate with cobalt bis-dicarbollide as a potential boron transporter for the tasks of boron neutron-capture therapy.
Materials and Methods. The experiments were carried out on Balb/c mice with induced murine colon carcinoma CT-26. Amino-amide chlorin e6 derivative conjugate with cobalt bis-dicarbollide was administered intravenously, the dose being 5 and 10 mg/kg body mass. The sampling for microscopic study of the drug uptake in ex vivo organs and tissues was performed 3 h after the administration.
Results. Characteristic nanoconjugate fluorescent peak was found in most organs under study, significant selectivity of the compound being noticed. A high uptake level was recorded in the liver, spleen and lung tissue. At the dose of 5 mg/kg, the drug content in a tumor was not different from that in muscular tissue and skin; maximum uptake was found in the liver. If the dose was increased up to 10 mg/kg, the nanoconjugate content in a tumor appeared to be comparable with that in the liver, the tumor/muscle ratio of fluorescent signals was ~3.
Conclusion. The study showed the prospects for using photosensitizer conjugate (chlorin е6) with boron particles as a means to deliver boron into a tumor. The level of the preparation uptake in tumor tissue depends on a dose.
Boron neutron capture therapy (BNCT) is a radiotherapy technique for malignancies characterized by high biological efficiency due to target radiation energy delivered directly to tumor cells. The method is based on relatively selective uptake of a non-radioactive isotope 10В in tumor cells and its further activation by a flux of thermal neutrons [1, 2]. Despite a high efficiency of the technique experimentally and clinically proved [3], up to the present, BNCT still cannot overstep the framework of clinical trials. There are several reasons for that, and one of them is the absence of effective techniques for boron delivery to tumor cells. The only approved compounds (3-(p-Boronophenyl)alanine: BPA, and sodium mercaptoundecahydro-closo-dodecaborate: BSH) approved for clinical use do not exhibit high uptake selectivity in tumor tissue [4–7].
To solve the problem of targeted boron delivery to tumor cells, there have been suggested different synthesis strategies of boron-based compound conjugates with bioactive molecules able to accumulate in tumor cells (precursors of nucleic acids, amino acids and peptides, carbohydrates, acridines, different polyamines, as well as porphyrins and phthalocyanines) [1, 2, 8, 9]. One of the most promising concepts in the sphere of preparations produced for BNCT is the use of porphyrin conjugates with polyhedral boron compounds. Oenbrink et al. were the first to study borated porphyrins for BNCT in 1988. Porphyrins and their boron derivatives were found to be able to accumulate in many solid tumors, they being two-in-one preparations: both boron delivery agents, and photosensitizers for photodynamic therapy [10]. The compounds of this category are bound through linker chlorin and boron polyhedron [11]. Representatives of such compounds effectively accumulate in tumor cells, while the use of carboranes as a boron element in a molecule enables an increase in the isotope concentration in a tumor and provides the boron moiety stability [12].
The essential stage preceding clinical trials is the study of pharmacokinetics of boron-based porphyrins, the assessment of their ability to accumulate and stay in a tumor at the higher concentration compared to healthy tissues, and then gets out of the body. Since the isotope 10B is non-radioactive, it can be neither used as a radioactive label, nor detected using appropriate techniques. In this case the key advantage of boron-based porphyrins is their ability to fluoresce that significantly facilitates the analysis of the preparation location and distribution in cells and living organisms using ex vivo and in vivo fluorescent imaging [13, 14]. These techniques enable the study of the fluorophore biodistribution in the body and assess its uptake in a tumor and healthy tissues.
The aim of the investigation was to study the biodistribution of amino-amide chlorin e6 derivative conjugate with cobalt bis-dicarbollide as a potential boron transfer in order to support boron neutron-capture therapy.
Materials and Methods
The compound under study, amino-amide chlorin e6 derivative conjugate with cobalt bis-dicarbollide (aminoalkyl-amide liker length, n=6) (Figure 1 (а)) was synthesized according to the technique previously developed [15].
The absorption and fluorescent spectra of a conjugate were assessed by a spectrophotometer-spectrofluorometer Synergy MX (BD, USA).
Animals and a tumor model. In vivo experiments were carried out in full agreement with the ethic principles established by European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (adopted in 18.03.1986 in Strasburg and approved in 15.06.2006 in Strasburg) and approved by the Ethics Committee of Nizhny Novgorod State Medical Academy. The biodistribution of boron-based conjugate was analyzed in Balb/c mice (female, 18–20 g, n=9) supplied by the laboratory mouse facility of the Branch of Institute of Bioorganic Chemistry, Russian Academy of Sciences (Pushchino, Moscow region). The animals were kept in standard conditions of vivarium with a 12-hour light day.
For experimental tumors we used murine colon carcinoma CT-26 (No.CRL-2638 according to ATCC® catalogue). The cells were cultured in DMEM (PanEco, Russia) containing 2 mM L-glutamine (PanEco, Russia) and 10% fetal calf serum (HyClone, USA), at 37°С and 5% СО2, in culture bottles, 75 cm2 in area. At each passage stage the cells were treated with 0.25% solution of trypsin and versene (PanEco, Russia). Subculturing was performed 2–3 days later, when the culture confluence reached 80% [16].
To induce tumors the animals (n=9) were injected with 1 million CT-26 cells in PBS, 100 µl, 10 mM (PanEco, Russia) subcutaneously in the thigh. The experiment was started on day 9–10 after the injection, when the tumor node diameter reached 9 mm.
In the study we used an initial solution of conjugate in cremophore, the active agent concentration being 2 mg/ml, which was diluted by physiological saline up to the concentration of 1 mg/ml. The control group (n=3) was not given the preparation. Three mice were injected with the conjugate using a dose of 5 mg/kg body mass, and three mice were given 10 mg/kg. The solution was injected via the caudal vein, the injection volume being 100 µl. 3 h after the boron-based conjugate injection the animals were sacrificed by the cervical vertebrae dislocation. For further microscopic study we used the samples of the following organs and tissues: tumors, striated muscles, skin, liver, kidney, spleen, small intestine, lungs.
Sample preparation and microscopic study. An entire tumor was cut from the surrounding tissue and dissected along a longitudinal axis perpendicular to the thigh surface. The femoral muscle was cut along fibers, and the kidney was cut along the sagittal plane of the organ. The liver, lung and spleen were sectioned deep longitudinally. The samples were analyzed immediately after they were prepared, without being fixed or frozen. Throughout the investigation the humidity of the preparations was maintained by moistening them with physiological saline.
The images were taken using a confocal laser scanning microscope Axiovert 200M LSM510 META (Carl Zeiss, Germany) with oil-immersion lens Plan-Apochromat, ×40, with numerical aperture 1.3. Lateral resolution of the images was 1024×1024 pixels, pixel size being 0.22 µm. The conjugate fluorescence was excited at the wavelength of 514 nm, a signal was recorded using a spectral module META in the 560–710 nm range at a pitch of 10 nm. Spectral information enabled one to identify a nanoconjugate signal against the background of tissue autofluorescence. All the imaged were recorded under identical conditions.
Data statistical processing. Using the microscope software, from the obtained spectral image we selected a channel that corresponded to the fluorescent signal in the 650–693 nm range. Using the program ImageJ (version 1.47v) we determined an average signal level over the each imaged area of the studied samples of tumor and healthy tissues. For each healthy tissue sample we studied from 3 to 5 fields of view. When analyzing tumor tissue samples the fields of view were randomly selected from central and peripheral parts of a tumor node (at least 5 fields of view for each sample). The data were presented as a mean signal value in arbitrary units ± a standard deviation for small samplings. For value comparison we used a one-way ANOVA test and Dunnett test.
Results and Discussion. The first stage of the investigation was to study optical properties of the obtained amino-amide chlorin e6 derivative conjugate with cobalt bis-dicarbollide. The compound was characterized by intense light absorption in a visible spectrum with the peaks at 405, 500 and 665 nm that were close to chlorin е6 peaks (Figure 1 (b)). Maximum fluorescence was found at 670 nm. The previous experiments on cell cultures [14] revealed an intense accumulation of the conjugate in А549 line tumor cells, which was characterized with a cell/medium distribution factor of ≈80. The property enables one to suggest the possibility to achieve a therapeutic dose necessary for BNCT, with higher contrast to healthy tissue and lower total toxicity in vivo. In vitro findings served as the basis to initiate the study on in vivo tumor models. The selection of experimental model CT-26 was due to high vascularization of a growing tumor [17].
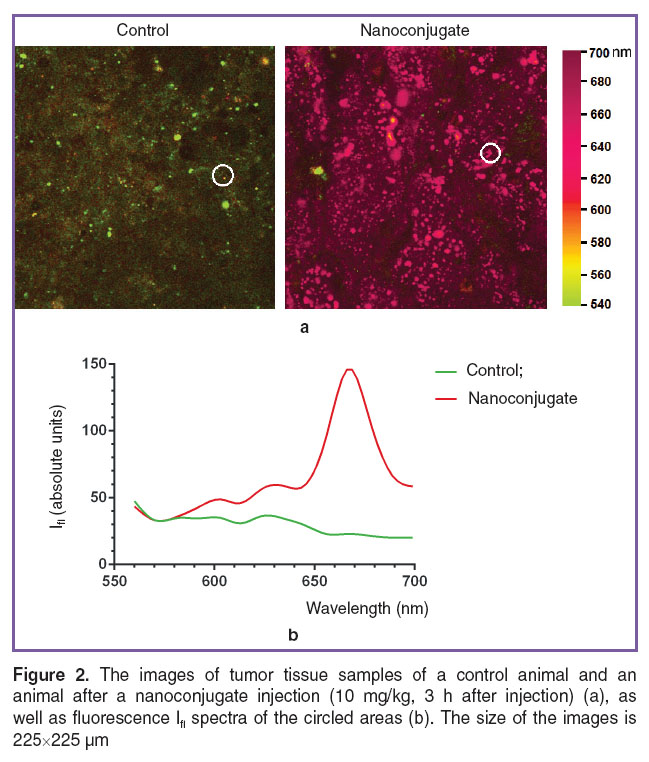
The study of tumor tissue samples revealed an intense fluorescent signal in it (Figure 2 (a)) at both nanoconjugate doses 3 h after the injection. Maximum fluorescence wasfound at 665–670 nm (Figure 2 (b)), which corresponded to spectrum of the conjugate under study.
The specific characteristic of fluorophore uptake in a tumor was its non-uniformity in different tumor parts (Figure 3). One might assume that the phenomenon is due to the so called EPR-effect (enhanced permeability and retention effect), i.e. the peculiarities of the tumor vasculature including the non-uniformity and chaotic state of forming vessels [18].
Characteristic fluorescence peak of the conjugate was found in most organs under study; in addition, the compound was characterized by significant uptake selectivity (Figure 4).
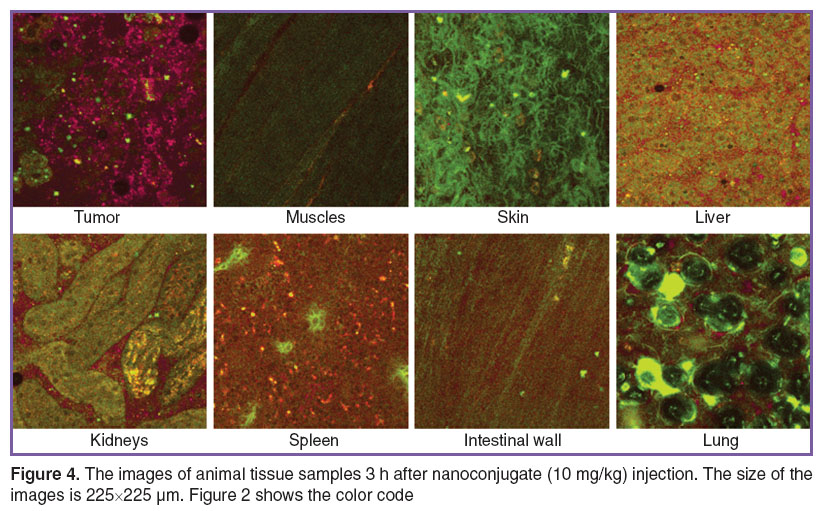 Figure 4. The images of animal tissue samples 3 h after nanoconjugate (10 mg/kg) injection. The size of the images is 225×225 µm. Figure 2 shows the color code Figure 4. The images of animal tissue samples 3 h after nanoconjugate (10 mg/kg) injection. The size of the images is 225×225 µm. Figure 2 shows the color code
|
High conjugate uptake level was recorded in the liver, kidneys, spleen and lung tissue that is consistent with the pharmacokinetics data received for preparations of chlorin family, and associated with morphofunctional characteristics of these organs [19]. A conjugate signal in skeletal muscles, as well as in muscular walls of hollow organs (stomach, intestines, heart), was significantly lower. Moreover, skin samples demonstrated nearly total absence of conjugate uptake. 3 h after the injection, a considerable amount of the substance remained in the circulation and was recorded in major vessels of different organs (Figure 4 shows the vessels feeding the skeletal muscle).
A quantitative analysis of the fluorescent signal level in tissue samples enabled one to reveal the dependence of nanoconjugate uptake on a preparation dose. After the conjugate injection, 5 mg/kg, its content in a tumor differed slightly from that in muscular tissue and skin, its maximum uptake being found in the liver (Figure 5).
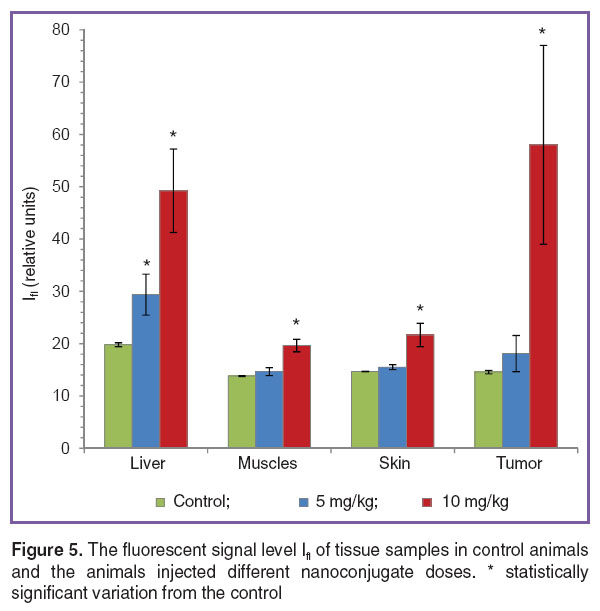 Figure 5. The fluorescent signal level Ifl of tissue samples in control animals and the animals injected different nanoconjugate doses. * statistically significant variation from the control Figure 5. The fluorescent signal level Ifl of tissue samples in control animals and the animals injected different nanoconjugate doses. * statistically significant variation from the control
|
If a dose is increased up to 10 mg/kg, the nanoconjugate content in a tumor appeared to be compatible with that in the liver, and the tumor/muscle ratio for fluorescent signals was ~3. It should be mentioned that the standard deviation of a signal level in a tumor was high if the nanoconjugate was injected at the dose of 10 mg/kg that was consistent with the microscopic study findings, when the tumor was found to have both the areas with high and average signal levels (See Figure 3).
Thus, the preliminary study showed a boron-based conjugate of chlorin to meet the requirements for preparations used for BNCT [2]. In particular, the compound demonstrated its ability to relatively selective uptake in a tumor, the “tumor/healthy tissue” ratio being 3:1. The conjugate exhibited low uptake in skin that enables one to expect its low phototoxicity and safety when used in the light. Chlorin property to fluoresce enables to control the boron content in tissues using in vivo techniques. These facts enable one to conclude the application perspectiveness of boron-based conjugates as the agents to deliver boron in BNCT. The next stage of the work will be the study of conjugate pharmacokinetics, as well as the analysis of boron content in a tumor and healthy tissues in order to develop optimal conditions and further radiation exposure.
Conclusion. The study of biodistribution of a photosensitizer conjugate (chlorin e6) with cobalt bis-dicarbollide demonstrated the perspectiveness of its application as a boron delivery means to a tumor. The uptake level of a conjugate in a tumor tissue depends on the dose administered.
Study Funding. This study was supported by a grant Russian Foundation for Basic Research No.14-02-00715.
Conflicts Interest. The authors have no conflicts of interest to disclose.
References
- Sivaev I.B., Bregadze V.I., Kuznetsov N.T. Derivatives of the closo-dodecaborate anion and their application in medicine. Russian Chemical Bulletin 2002; 51(8): 1362–1374, http://dx.doi.org/10.1023/A:1020942418765.
- Rozumenko V.D. Boron neutron capture therapy of brain tumors. Ukrainskiy neyrokhirurgicheskiy zhurnal 2001; 3: 4–12.
- Hartman T., Carlson J. Radiation dose heterogeneity in receptor and antigen mediated boron neutron capture therapy. Radiother Oncol 1994; 31(1): 61–75, http://dx.doi.org/10.1016/0167-8140(94)90414-6.
- Boron chemistry at the beginning of the 21st century. Bubnov Yu.N. (editor). Moscow: Editorial URSS; 2003; 376 p.
- Soloway A.H., Tjarks W., Barnum B.A., Rong F.G., Barth R.F., Codogni I.M., Wilson J.G. The chemistry of neutron-capture therapy. Chem Rev 1998; 98(4): 1515–1562, http://dx.doi.org/10.1021/cr941195u.
- Yokoyama K., Miyatake S., Kajimoto Y., Kawabata S., Doi A., Yoshida T., Asano T., Kirihata M., Ono K., Kuroiwa T. Pharmacokinetic study of BSH and BPA in simultaneous use for BNCT. J Neurooncol 2006; 78(3): 227–232, http://dx.doi.org/10.1007/s11060-005-9099-4.
- Goudgaon N.M., El-Kattan G.F., Schinazi R.F. Boron containing pyrimidines, nucleosides, and oligonucleotides for neutron capture therapy. Nucleosides and Nucleotides 1994; 13(1–3): 849–880, http://dx.doi.org/10.1080/15257779408013283.
- Neutron capture therapy — principles and application. Sauerwein W., Wittig A., Moss R., Nakagawa Y. (editors). Springer Science + Business Media; 2012, http://dx.doi.org/10.1007/978-3-642-31334-9.
- Yamamoto Y., Cai J., Nakamura H., Sadayori N., Asao N., Nemoto H. Synthesis of netropsin and distamycin analogues bearing o-carborane and their DNA recognition. Journal of Organic Chemistry 1995; 60(11): 3352–3357, http://dx.doi.org/10.1021/jo00116a018.
- Oenbrink G., Jurgenlimke P., Gabel D. Accumulation of porphyrins in cells influence of hydrophobicity aggregation and protein binding. Photochem Photobiol 1988; 48(4): 451–456, http://dx.doi.org/10.1111/j.1751-1097.1988.tb02844.x.
- Grin M.A., Titeev R.A., Brittal D.I., Chestnova A.V., Mironov A.F., Feofanov A.V., Lobanova I.A., Sivaev I.B., Bregadze V.I. Synthesis of cobalt bis(dicarbollide) conjugates with natural chlorins by the Sonogashira reaction. Russian Chemical Bulletin 2010; 59(1): 219–224, http://dx.doi.org/10.1007/s11172-010-0065-8.
- Efremenko A.V., Ignatova A.A., Grin M.A., Mironov A.F., Bregadze V.I., Sivaev I.B., Feofanov A.V. Confocal microscopy and spectral imaging technique: contribution to the development of neutron sensitizers for anticancer BNCT. In: Current microscopy contributions to advances in science and technology. A. Méndez-Vilas (editor). Spain: FORMATEX, 2012; p. 84–90.
- Ol'shevskaya V.A., Zaytsev A.V., Savchenko A.N., Shtil A.A., Cheong C.S., Kalinin V.N. Boronated porphyrins and chlorins as potential anticancer drugs. Bulletin of the Korean Chemical Society 2007; 28(11): 1910–1914.
- Efremenko A.V., Ignatova A.A., Grin M.A., Sivaev I.B., Mironov A.F., Bregadze V.I., Feofanov A.V. Chlorin e6 fused with a cobalt-bis(dicarbollide) nanoparticle provides efficient boron delivery and photoinduced cytotoxicity in cancer cells. Photochem Photobiol Sci 2014; 13(1): 92–102, http://dx.doi.org/10.1039/c3pp50226k.
- Grin M.A., Titeev R.A., Brittal D.I., Ulybina O.V., Tsiprovskiy A.G., Berzina M. Ya., Lobanova I.A., Sivaev I.B., Bregadze V.I., Mironov A.F. New conjugates of cobalt bis(dicarbollide) with chlorophyll a derivatives. Mendeleev Communications 2011; 21(2): 84–86, http://dx.doi.org/10.1016/j.mencom.2011.03.008.
- Freshni R.Ya. Kul’tura zhivotnykh kletok: prakticheskoe rukovodstvo [Culture of animal cells: practice guidelines]. Moscow: BINOM. Laboratoriya znaniy; 2010; 691 p.
- Kuznetsov S.S., Snopova L.B., Karabut М.М., Sirotkina М.А., Buyanova N.L., Kalganova Т.I., Elagin V.V., Senina-Volzhskaya I.V., Barbashova L.N., Shumilova A.V., Zagaynova E.V., Vitkin A., Gladkova N.D. Features of morphological changes in experimental ct-26 tumors growth. Sovremennye tehnologii v medicine 2015; 7(3): 32–39, http://dx.doi.org/10.17691/stm2015.7.3.04.
- Maeda H. Macromolecular therapeutics in cancer treatment: the EPR effect and beyond. Journal of Controlled Release 2012; 164(2): 138–144, http://dx.doi.org/10.1016/j.jconrel.2012.04.038.
- Juzeniene A. Chlorin e6-based photosensitizers for photodynamic therapy and photodiagnosis. Photodiagnosis Photodyn Ther 2009; 6(2): 94–96, http://dx.doi.org/10.1016/j.pdpdt.2009.06.001.