A New Quantitative Method for Assessing the Ultrastructure of Cardiomyocyte Mitochondria of the Right Atrial Appendage
The aim of this study was to develop a quantitative method for assessing the ultrastructure of cardiomyocyte mitochondria in the right atrial appendage of patients with chronic heart failure (CHF).
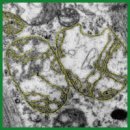
Materials and Methods. A single-center prospective study included 39 patients aged 67 [58; 71] years who underwent coronary artery bypass grafting. The inclusion criteria were as follows: heart failure with a left ventricular ejection fraction of <50%, atherosclerotic plaques in two or more large coronary arteries of 70% or greater, and the cardiac team’s decision to perform coronary artery bypass grafting. To perform electron microscopy, biopsy samples were collected from the right atrial appendage during coronary artery bypass grafting. Two calculated parameters were used for the analysis: “total area of interfibrillar mitochondria” and “ratio of outer to inner membrane length”. Based on these, the total index was calculated by the formula: the “total area of interfibrillar mitochondria” / the “ratio of outer to inner membrane length”.
Results. The median values of the calculated parameters characterizing the ultrastructure of cardiomyocyte mitochondria in the right atrial appendage were as follows: 43.7 [35.9; 54.3]% for the “total area of interfibrillar mitochondria”, 31 [25; 37]% for the “ratio of outer to inner membrane length”, and 1.4 [0.95; 2.00] for the total index accounting for both ultrastructural characteristics of mitochondria: the “total area of interfibrillar mitochondria” / the “ratio of outer to inner membrane length”.
To assess the clinical significance of the proposed total index, associations between the obtained values and atrial fibrillation (AF) were studied. Mitochondrial ultrastructural changes were more pronounced in patients with both AF and CHF. However, some parameters had no statistically significant differences: the total area of interfibrillar mitochondria was lower in patients with AF (42% vs 49%, p=0.224), and the ratio of outer to inner membrane length was higher in patients with AF (35% vs 31%, p=0.125). The total index was significantly lower in patients with AF (0.96 vs 1.75, p=0.021). ROC analysis was performed to identify a connection between total index and AF, and AUC was 0.773 (p=0.021).
Conclusion. The proposed total index for analyzing cardiomyocyte micrographs, calculated as the “total area of interfibrillar mitochondria” / the “ratio of outer to inner membrane length”, provides a comprehensive characterization of mitochondria that accounts not only for their number and size but also for their internal structure.
- Galyavich A.S., Tereshchenko S.N., Uskach T.M., Ageev F.T., Aronov D.M., Arutyunov G.P., Begrambekova Yu.L., Belenkov Yu.N., Boytsov S.A., Bubnova M.G., Vasyuk Yu.A., Villevalde S.V., Vinogradova N.G., Garganeeva A.A., Gendlin G.E., Gilyarevsky S.R., Glezer M.G., Gautier S.V., Grinstein Yu.I., Dovzhenko T.V., Drapkina O.M., Duplyakov D.V., Zhirov I.V., Zateishchikov D.A., Zvartau N.E., Irtyuga O.B., Kobalava Zh.D., Koziolova N.A., Koroteev A.V., Libis R.A., Lopatin Yu.M., Mareev V.Yu., Mareev Yu.V., Matskeplishvili S.T., Mikhailov E.N., Nasonova S.N., Narusov O.Yu., Nedogoda S.V., Nedoshivin A.O., Ovchinnikov A.G., Orlova Ya.A., Perepech N.B., Pogosova N.V., Rimskaya E.M., Samko A.N., Saidova M.A., Sapelnikov O.V., Safiullina A.A., Sitnikova M. Yu., Skvortsov A.A., Skibitskiy V.V., Stukalova O.V., Tarlovskaya E.I., Tereshchenko A.S., Chesnikova A.I., Fedotov P.A., Fomin I.V., Khasanov N.R., Shevchenko A.O., Shaposhnik I.I., Shariya M.A., Shlyakhto E.V., Yavelov I.S., Yakushin S.S. 2024 clinical practice guidelines for chronic heart failure. Russian Journal of Cardiology 2024; 29(11): 6162, https://doi.org/10.15829/1560-4071-2024-6162.
- Paillard M., Abdellatif M., Andreadou I., Bär C., Bertrand L., Brundel B.J.J.M., Chiva-Blanch G., Davidson S.M., Dawson D., Di Lisa F., Evans P., Giricz Z., Hausenloy D.J., Kleinbongard P., Lezoualc'h F., Liehn E., Maack C., Maguy A., Murphy E., Perrino C., Pesce M., Rainer P.P., Streckfuss-Bömeke K., Thielmann M., Tian R., Tocchetti C.G., Van Der Velden J., Van Linthout S., Zacchigna S., Krieg T. Mitochondrial targets in ischaemic heart disease and heart failure, and their potential for a more efficient clinical translation. A scientific statement of the ESC Working Group on Cellular Biology of the Heart and the ESC Working Group on Myocardial Function. Eur J Heart Fail 2025; 27(9): 1720–1736, https://doi.org/10.1002/ejhf.3674.
- Garganeeva A.A., Tukish O.V., Kuzheleva E.A., Muslimova E.F., Gulya M.O., Zhargasova V.A., Popov S.V. Heart failure and mitochondrial dysfunction: research methods in experiment and clinical practice. Acta Biomedica Scientifica 2025; 10(1): 103–114, https://doi.org/10.29413/ABS.2025-10.1.11.
- Stepanov A.V., Baidyuk E.V., Sakuta G.A. The features of mitochondria of cardiomyocytes from rats with chronic heart failure. Cell and Tissue Biology 2017; 11(6): 458–465, https://doi.org/10.1134/s1990519x17060086.
- Kuzheleva E.A., Garganeeva A.A., Tukish O.V., Vitt K.N., Andreev S.L., Syromyatnikova E.E., Vladimirova E.A. Total area of interfibrillary mitochondria in cardiomyocytes of the right atrial appendix as an indicator of the functional state of the cardiovascular system in chronic heart failure. Bulletin of Experimental Biology and Medicine 2024; 178(10): 497–502, https://doi.org/10.47056/0365-9615-2024-178-10-497-502.
- El'darov C.M., Vays V.B., Vangeli I.M., Bakeeva L.E., Kolosova N.G. Morphometric examination of mitochondrial ultrastructure in aging cardiomyocytes. Biochemistry 2015; 80(5): 604–609, https://doi.org/10.1134/s0006297915050132.
- Garganeeva A.A., Kuzheleva E.A., Tukish O.V., Vitt K.N., Andreev S.L., Muslimova E.F., Korepanov V.A., Afanasiev S.A., Gulya M.O., Syromyatnikova E.E., Vladimirova E.A., Stepanov I.V. Possibilities of mitochondrial dysfunction diagnosis in chronic heart failure. Siberian Journal of Clinical and Experimental Medicine 2024; 39(3): 51–57, https://doi.org/10.29001/2073-8552-2024-39-3-51-57.
- McDonagh T.A., Metra M., Adamo M., Gardner R.S., Baumbach A., Böhm M., Burri H., Butler J., Čelutkienė J., Chioncel O., Cleland J.G.F., Crespo-Leiro M.G., Farmakis D., Gilard M., Heymans S., Hoes A.W., Jaarsma T., Jankowska E.A., Lainscak M., Lam C.S.P., Lyon A.R., McMurray J.J.V., Mebazaa A., Mindham R., Muneretto C., Francesco Piepoli M., Price S., Rosano G.M.C., Ruschitzka F., Skibelund A.K.; ESC Scientific Document Group 2023. Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2023; 44(37): 3627–3639, https://doi.org/10.1093/eurheartj/ehad195.
- Dabravolski S.A., Zhuravlev A.D., Kartuesov A.G., Borisov E.E., Sukhorukov V.N., Orekhov A.N. Mitochondria-mediated cardiovascular benefits of sodium-glucose co-transporter 2 inhibitors. Int J Mol Sci 2022; 23(10): 5371, https://doi.org/10.3390/ijms23105371.
- Schwemmlein J., Maack C., Bertero E. Mitochondria as therapeutic targets in heart failure. Curr Heart Fail Rep 2022; 19(2): 27–37, https://doi.org/10.1007/s11897-022-00539-0.
- Hayashi D., Ohshima S., Isobe S., Cheng X.W., Unno K., Funahashi H., Shinoda N., Okumura T., Hirashiki A., Kato K., Murohara T. Increased (99m)Tc-sestamibi washout reflects impaired myocardial contractile and relaxation reserve during dobutamine stress due to mitochondrial dysfunction in dilated cardiomyopathy patients. J Am Coll Cardiol 2013; 61(19): 2007–2017, https://doi.org/10.1016/j.jacc.2013.01.074.
- Bidaoui G., Assaf A., Marrouche N. Atrial fibrillation in heart failure: novel insights, challenges, and treatment opportunities. Curr Heart Fail Rep 2024; 22(1): 3, https://doi.org/10.1007/s11897-024-00691-9.
- Carlisle M.A., Fudim M., DeVore A.D., Piccini J.P. Heart failure and atrial fibrillation, like fire and fury. JACC Heart Fail 2019; 7(6): 447–456, https://doi.org/10.1016/j.jchf.2019.03.005.
- Zdebik N., Poręba R., Gać P. Importance of T1-mapping sequence in patients with hypertrophic cardiomyopathy without foci of non-ischemic myocardial injury in late gadolinium enhancement sequence. Biomedicines 2024; 12(6): 1330, https://doi.org/10.3390/biomedicines12061330.
- Sukhacheva T.V., Vaskovsky V.A., Revishvili A.Sh., Serov R.A. Morphological features of the myocardium of the atrial appendages in patients with different forms of atrial fibrillation. Arkhiv patologii 2017; 79(4): 3–12, https://doi.org/10.17116/patol20177943-12.
- Guttipatti P., Saadallah N., Ji R., Avula U.M.R., Goulbourne C.N., Wan E.Y. Quantitative 3D electron microscopy characterization of mitochondrial structure, mitophagy, and organelle interactions in murine atrial fibrillation. J Struct Biol 2024; 216(3): 108110, https://doi.org/10.1016/j.jsb.2024.108110.
- Qi X., Xiong F., Xiao J., Muthukumarasamy K.M., Altuntas Y., Zhong Y., Abu-Taha I., Bruns F., Tekook M., Kamler M., Villeneuve L., Nozza A., Sirois M., Karch J., Pasdois P., Bers D.M., Dobrev D., Nattel S. Time-dependent mitochondrial remodeling in experimental atrial fibrillation and potential therapeutic relevance. Preprint. bioRxiv 2025; 2025.01.29.635508, https://doi.org/10.1101/2025.01.29.635508.
- Paillard M., Abdellatif M., Andreadou I., Bär C., Bertrand L., Brundel B.J.J.M., Chiva-Blanch G., Davidson S.M., Dawson D., Di Lisa F., Evans P., Giricz Z., Hausenloy D.J., Kleinbongard P., Lezoualc'h F., Liehn E., Maack C., Maguy A., Murphy E., Perrino C., Pesce M., Rainer P.P., Streckfuss-Bömeke K., Thielmann M., Tian R., Tocchetti C.G., Van Der Velden J., Van Linthout S., Zacchigna S., Krieg T. Mitochondrial targets in ischaemic heart disease and heart failure, and their potential for a more efficient clinical translation. A scientific statement of the ESC Working Group on Cellular Biology of the Heart and the ESC Working Group on Myocardial Function. Eur J Heart Fail 2025; 27(9): 1720–1736, https://doi.org/10.1002/ejhf.3674.










